Authority for the officers of Drug Good Manufacturing Practice (GMP) Inspectorate Task Force to conduct inspection and post marketing surveillance in accordance with Republic Act 9711, even beyond office hours, as the need arises in Luzon, Visayas and Mindanao from January to December 2019

FDA PERSONNEL ORDER No. 2019-0020 SUBJECT: Authority for the officers of Drug Good Manufacturing Practice (GMP) Inspectorate Task Force to conduct inspection and post marketing surveillance in accordance with Republic […]
FDA Advisory No. 2018-327-A || Amendment to FDA Advisory No. 2018-327 “Public Health Warning Against the Use of Unnofified Cosmetic Products (Batch 16)” Dated December 2018
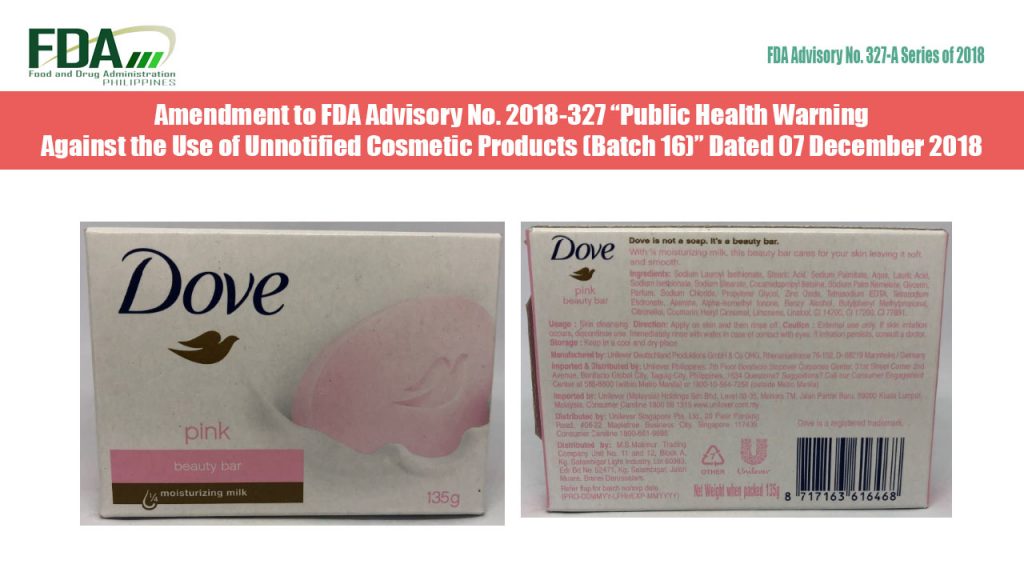
The Food and Drug Administration (FDA) hereby advises the public that the list of unnotified cosmetic products listed in FDA Advisory No. 2018-327 “Public Health Warning Against the Use of […]
FDA Advisory No. 2019-001 || Public Health Warning Against the Purchase and Use of Unregistered Medical Device “Baby One Digital Thermometer”
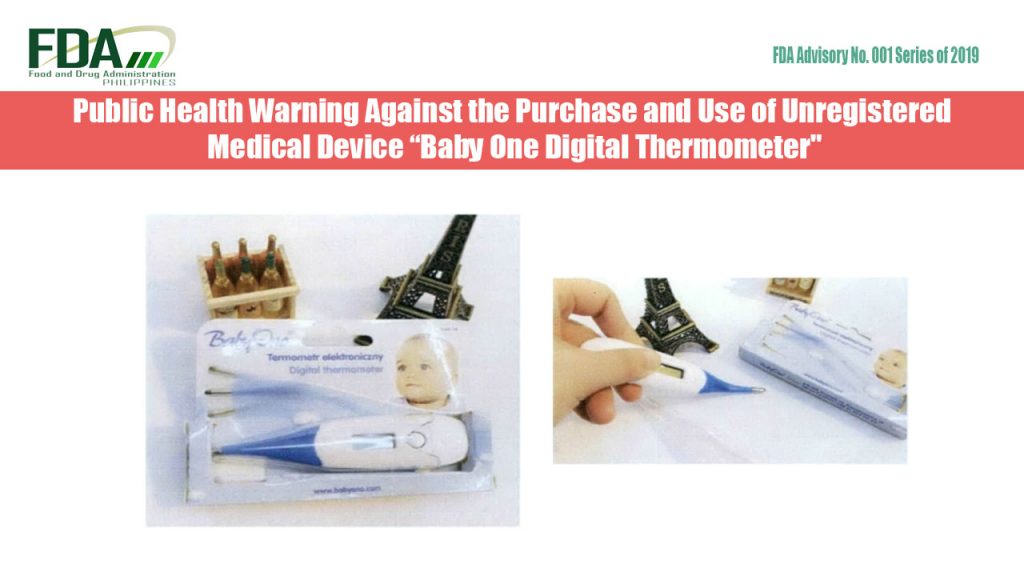
to continue reading, click attachment below… FDA Advisory No. 2019-001



