FDA Advisory No. 2019-130-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:
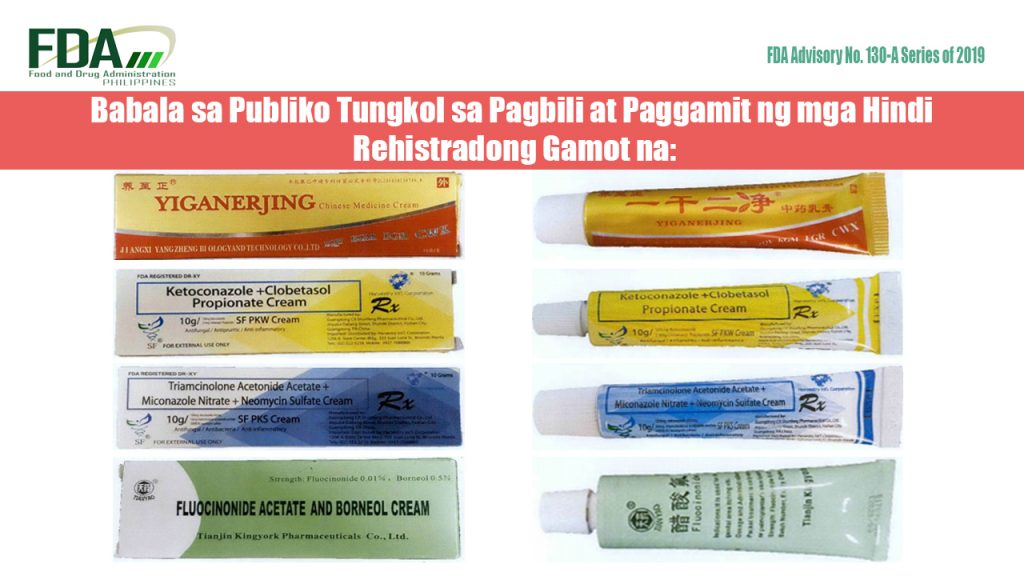
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga rehistradong gamot na: 1. YIGANERJING Chinese Medicine Cream 2. Ketoconazole + Clobetasol Propionate Cream […]
FDA Advisory No. 2019-130 || Public Health Warning Against the Purchase and Use of the following Unregistered Drug Products

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: YIGANERJING Chinese Medicine Cream Ketoconazole + Clobetasol Propionate Cream SF […]
FDA Advisory No. 2019-129 || FDA Email Directory

To ensure ease of transaction and the desire to provide a more efficient customer service, the Food and Drug Administration (FDA) hereby advises the public to send specific concerns, queries, […]
FDA Advisory No. 2018-327-C || Amendment to FDA Advisory No. 2018-327 “Public Health Warning Against the Use of Unnotified Cosmetic Products (Batch 16)” Dated 07 December 2018

The Food and Drug Administration (FDA) hereby advises the public that the list of unnotified cosmetic products listed in FDA Advisory No. 2018-327 ” Public Health Warning Against the Use […]
FDA Advisory No. 2019-128 || Safe Information: Fluoroquinolones and risk of Aortic Aneurysm and Dissection

Introduction The US FDA searched their adverse event reporting system database and reviewed epidemiological studies to investigate the association between fluoroquinolone antibacterial use and the risk of aortic aneurysm and […]
FDA Advisory No. 2019-127 || Public Warning on the Purchase and Consumption of Processed Pork Meat Product from Identified Countries Suspected to be affected by African Swine fever virus

to continue reading, click the link below… FDA Advisory No. 2019-127
FDA Advisory No. 2019-126 || Public Health Warning Against the Purchase and Use of Counterfeit Cosmetic Products (Batch 7)

The Food and Drug Administration (FDA) hereby advises the public against the purchase and use of M·N® MENOW GENERATION-II LONG LASTING LIP GLOSS (VARIANT 34) whose particulars/details are provided below: […]
FDA Advisory No. 2019-125 || Public Health Warning Against the Purchase and Use of Counterfeit Cosmetic Products (Batch 6)

The Food and Drug Administration (FDA) hereby advises the public against the purchase and use of MAYBELLINE NEW YORK THE MAGNUM VOLUM’ EXPRESS WATERPROOF MASCARA whose particulars/details are provided below: PRODUCT DETAILS: […]
FDA Advisory No. 2019-124 || Public Health Warning Against the Purchase and Use of Counterfeit Cosmetic Products (Batch 5)

The Food and Drug Administration (FDA) hereby advises the public against the purchase and use of the following counterfeit cosmetic products whose particulars/details are provided in the attached annex: 3W […]
FDA Advisory No. 2019-123 || Public Health Warning Against the Purchase and Use of Counterfeit Cosmetic Products (Batch 4)
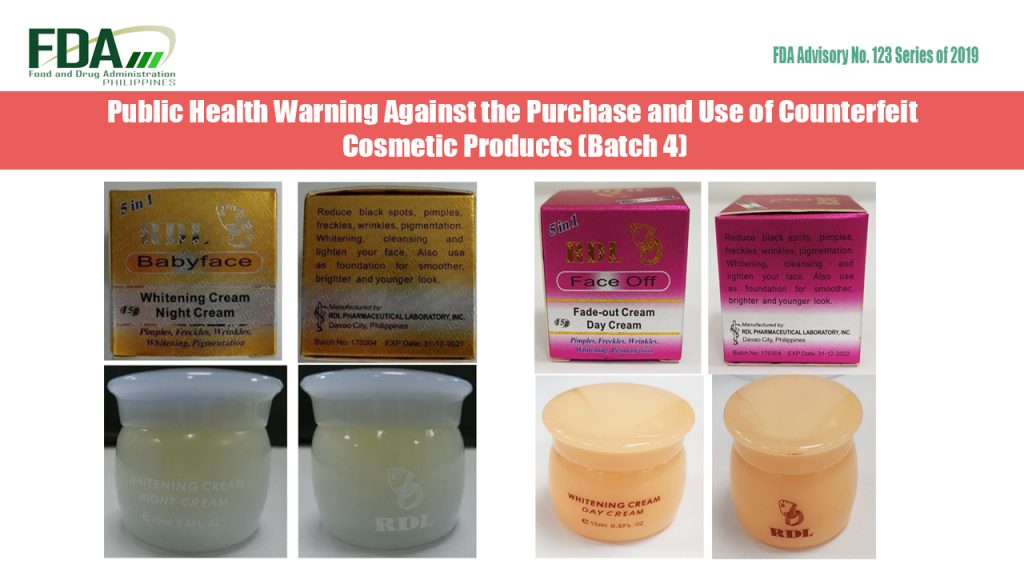
The Food and Drug Administration (FDA) hereby advises the public against the purchase and use of the following counterfeit cosmetic products whose particulars/details are provided in the attached annex: RDL […]



