FDA Advisory No. 2019-223 || Public Health Warning Against the Purchase and Consumption of the Following Unregistered Food Products and Food Supplements:

The Food and Drug Administration (FDA) warns the public from purchasing and consuming the following unregistered food products and food supplements: FRUCCY’S Ginger Tea GARDEN’S SECRET 16 in 1 Turmeric […]
FDA Advisory No. 2019-222 || Public Health Warning Against the Purchase and Consumption of the Following Unregistered Food Products:

The Food and Drug Administration (FDA) warns the public from purchasing and consuming the following unregistered food products: MUNCH A BUNCH Araro ARA’S MAMI HOUSE & RESTAURANT Gourmet Bangus Fresh […]
FDA Advisory No. 2019-221 || Public Health Warning Against the Purchase and Consumption of the Following Unregistered Food Products:

The Food and Drug Administration (FDA) warns the public from purchasing and consuming the following unregistered food products: MINI POPZA Lollipop Artificial Cola Flavoured MINI POPZA Lollipop Artificial Sprite Flavoured […]
FDA Advisory No. 2019-220 || Public Health Warning Against the Purchase and Consumption of the Following Unregistered Food Products:

The Food and Drug Administration (FDA) warns the public from purchasing and consuming the following unregistered food products: GREENFREE Orange & Menthol Flavoured Candy GREENFREE Blueberry & Menthol Flavoured Candy […]
FDA Advisory No. 2019-219 || Public Health Warning Against the Purchase and Consumption of the Following Unregistered Food Products:

The Food and Drug Administration (FDA) warns the public from purchasing and consuming the following unregistered food products: ZOOD ZOOD Bubble Gum Grape Flavoured ZOOD ZOOD Bubble Gum Orange Flavoured […]
ADDITIONAL TRAINING SCHEDULE OF FDA ACADEMY FOR 2019

Please be informed of the additional FDA Academy training schedule for 2019. COURSE TITLE DATE VENUE Center for Cosmetic Regulation and Research QPIRA Seminar 3-4 September 2019 NCR Unified Licensing […]
RESCHEDULE OF UNIFIED LICENSING SEMINAR FOR REGION II STAKEHOLDERS TO 7 AUGUST 2019

Please be informed that the Unified Licensing Seminar for Region II Stakeholders (Course Code:ULS-RII) in Isabela originally scheduled on 18 July 2019 shall be rescheduled on 7 August 2019. Seminar […]
FDA Advisory No. 2019-218 || Public Health Warning Against the Purchase and Use of the Unregistered Medical Device “INTROCAN® – W (NON- SAFETY)”
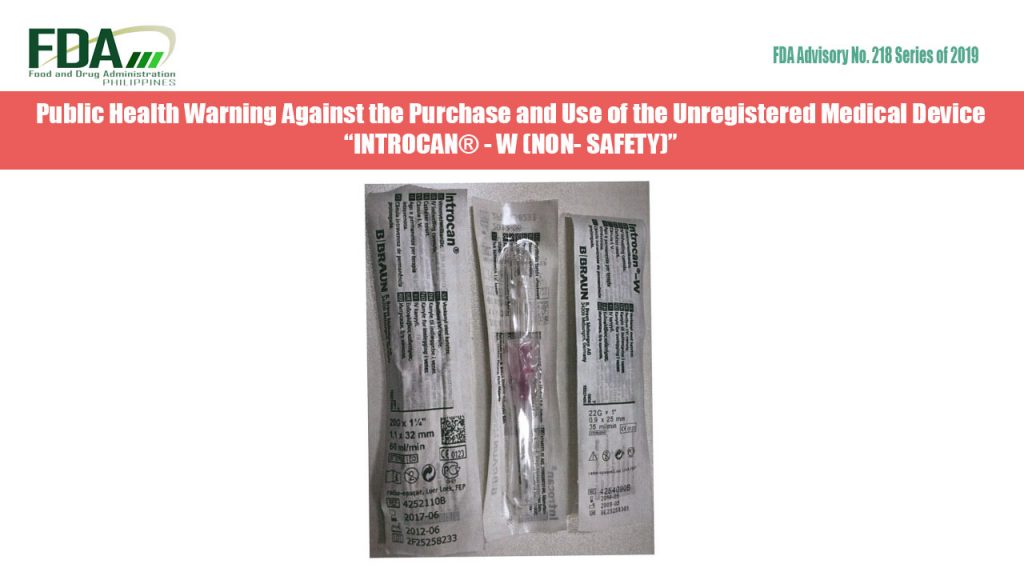
The Food and Drug Administration (FDA) warns all healthcare professionals and the general public against the purchase and use of the unregistered medical device: “INTROCAN® – W (NON- SAFETY)” The […]
FDA Advisory No. 2019-217 || Public Health Warning Against the Purchase and Use of the Unregistered Medical Device “KOJAK® SELINGE-SYRINGE WITH NEEDLE”
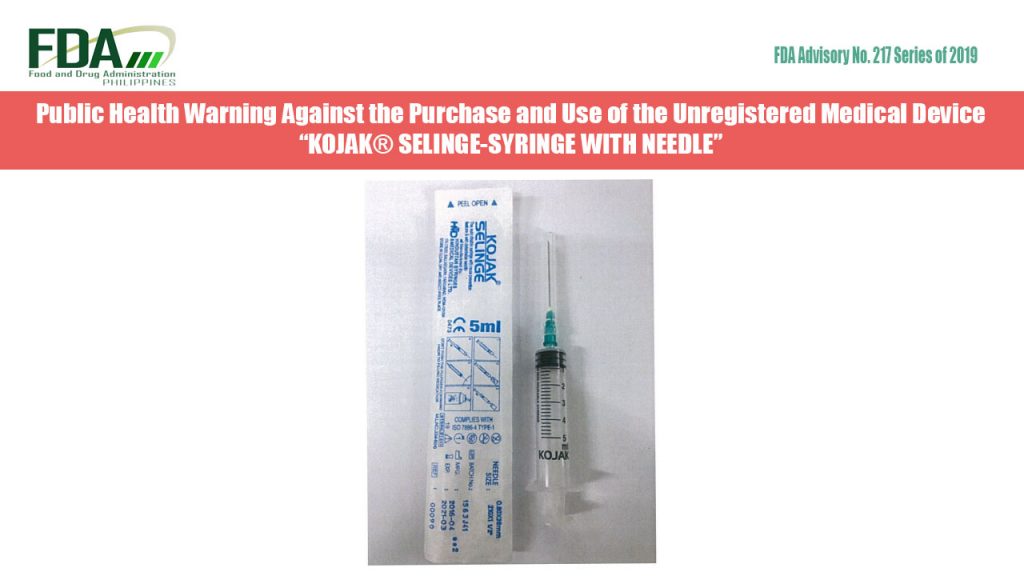
The Food and Drug Administration (FDA) warns all healthcare professionals and the general public against the purchase and use of the unregistered medical device: “Coralite® Cold-Wrap Elastic Bandage” The FDA […]
FDA Advisory No. 2019-216 || Public Health Warning Against the Purchase and Use of the Unregistered Medical Device “Coralite® Cold-Wrap Elastic Bandage”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public against the purchase and use of the unregistered medical device: “Coralite® Cold-Wrap Elastic Bandage” The FDA […]



