ANNOUNCEMENT || Launching of FDA VERIFICATION PORTAL

The Food and Drug Administration,in its commitment to provide stakeholders with streamlined and improved government services, has developed the FDA Verification Portal System. This online portal provides comprehensive lists of […]
FDA Advisory No. 2020-1688 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:
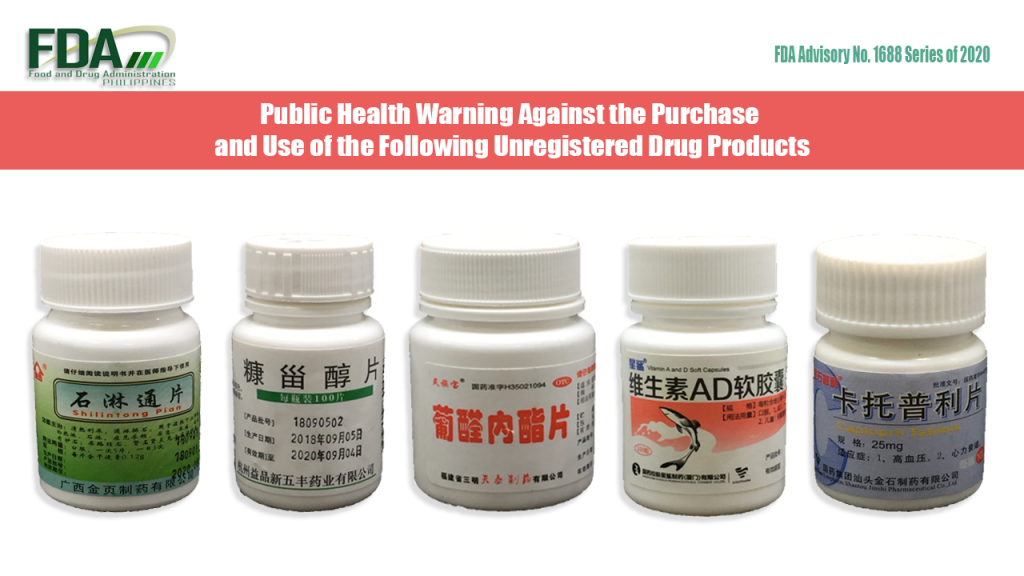
The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: Shilintong Pian Xinwufeng H33022029 [Label in Foreign Language] OTC H35021094 [Label in […]
FDA Advisory No. 2020-1687-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:
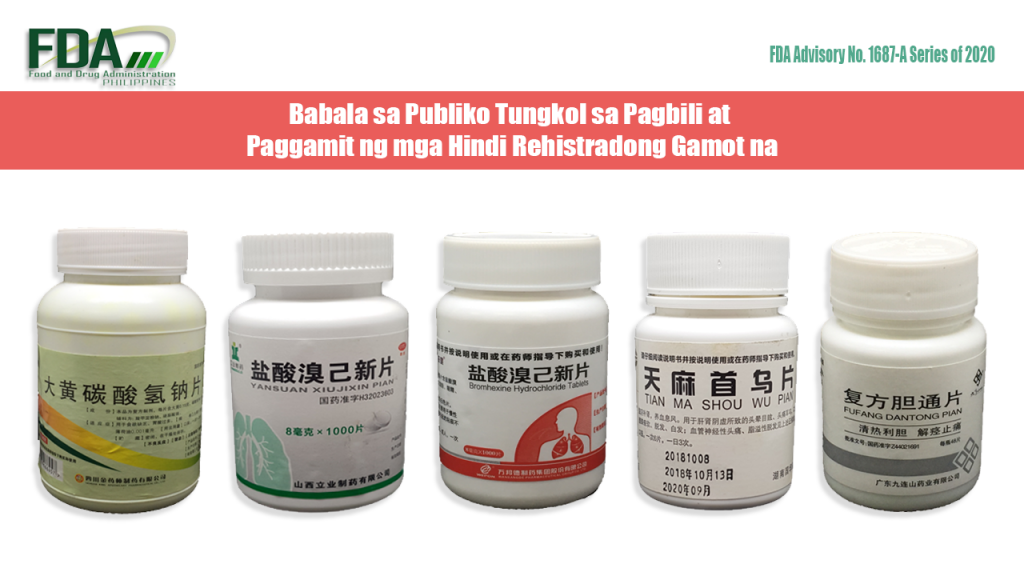
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: OTC H51021671 [Label in Foreign Language] OTC Yansuan Xiujixin Pian OTC Wepon® Bromhexine Hydrochloride […]
FDA has released a total of 260 COVID – 19 Test Kits (89 – PCR based, 93 – Rapid Antibody, 58 – Immunoassay and 20 – Others)

As of 29 September 2020, FDA has released a total of 260 COVID – 19 Test Kits (89 – PCR based, 93 – Rapid Antibody , 58 – Immunoassay and 20 […]
FDA Advisory No. 2020-1687 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:
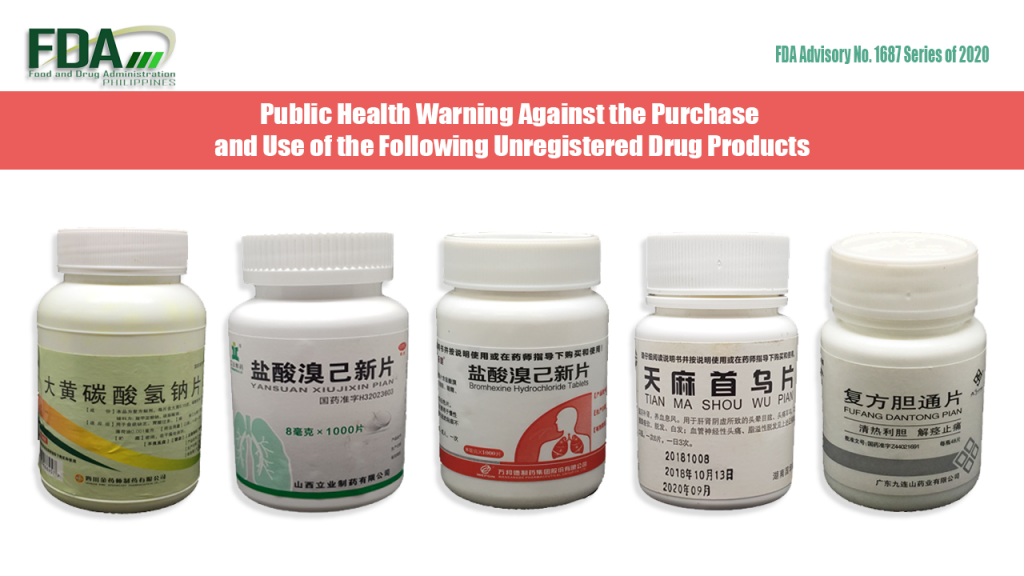
The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: OTC H51021671 [Label in Foreign Language] OTC Yansuan Xiujixin Pian OTC […]
FDA Advisory No. 2020-1686-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:
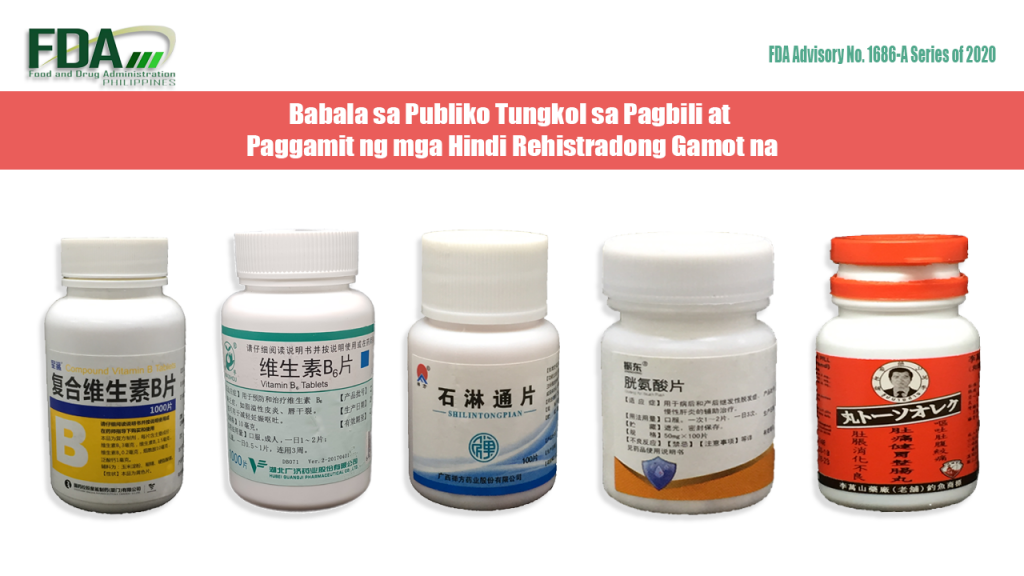
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: OTC Compound Vitamin B Tablets OTC Miaoshou® Vitamin B6 Tablets Shilintongpian Zhendong® Guang An Suan […]
FDA Advisory No. 2020-704-A || Lifting the Advisory on the Notified Product “CHICCO DENTIFRICIO TOOTHPASTE (MELA-APPLE BANANA)” under FDA Advisory No. 2020-704 “Public Health Warning Against the Purchase and Use of Unnotified Cosmetic CHICCO DENTIFRICIO TOOTHPASTE (MELA-APPLE BANANA)” Dated 16 April 2020

The Food and Drug Administration (FDA) informs the public that the Cosmetic product CHICCO DENTIFRICIO TOOTHPASTE (MELA-APPLE BANANA) with Notification No. 1000006556643 has been notified by the Market Authorization Holder, Europlay Distributor Company, Inc., […]
FDA Advisory No. 2020-1104-A || Lifting the Advisory on the Notified Cosmetic Product “MANUKA HEALTHY HONEY 100% NATURAL LIP BALM” under FDA Advisory No. 2020-1104 “Public Health Warning Against the Purchase and Use of Unauthorized Cosmetic Product MANUKA HEALTHY HONEY 100% NATURAL LIP BALM” Dated 16 June 2020

The Food and Drug Administration (FDA) informs the public that the Cosmetic product MANUKA HEALTHY HONEY 100% NATURAL LIP BALM with Notification No. 1000006589658 has been notified by the Market Authorization Holder, Bee Healthy Trading […]
ANNOUNCEMENT || RESCHEDULE OF ONLINE PUBLIC CONSULTATION ON THE DRAFT GUIDELINES FOR THE REGULATION OF VAPOR PRODUCTS AND HEATED TOBACCO PRODUCTS IN RELATION TO THE PROVISIONS UNDER RA 11346, RA 11467 AND EO 106

Due to unforeseen technical issues with the registration link wherein several stakeholders are unable to access and submit the online registration form, we would like to inform all concerned stakeholders […]
Administrative Order No. 2020-0044 || Adoption of the Collaborative Procedure for the Accelerated Registration of World of Health Organization (WHO) – Prequalified Pharmaceutical Products and Vaccines

Attachment:-> Administrative Order 2020-0044



