As a regulatory agency under the Department of Health, the Food and Drug Administration, created under Republic Act No. 3720, series of 1963, as amended by Executive Order 175, series of 1987, otherwise known as the “Food, Drugs and Devices, and Cosmetics Act”, and subsequently Republic Act No. 9711 otherwise known as “The Food and Drug Administration Act of 2009”, is mandated to ensure the safety, efficacy or quality of health products which include food, drugs, cosmetics, devices, biologicals, vaccines, in-vitro diagnostic reagents, radiation-emitting devices or equipment, and household/urban hazardous substances, including pesticides and toys, or consumer products that may have an effect on health which require regulations as determined by the FDA.
Among others, the FDA is also mandated to enforce the provisions of the following laws:
- RA
9502, or The Universally Accessible Cheaper and Quality Medicine Act of
2008
- RA 6675, or The Generics Act Of 1988,
- RA 10918, or The Pharmacy Law,
- RA 9211, or The Tobacco Regulation Act of 2003
- RA 7394, or The Consumer Act of the Philippines
- RA 7581/10623, or The Price Act
- RA 10611, or The Food Safety Act of 2013
- RA 8172, or The ASIN Law,
- RA 8203, or The Special Law on Counterfeit Drug
- RA 8976, or The Food Fortification Law
- RA 9165, or The Comprehensive Dangerous Drugs Act
- RA 9257, or The Expanded Senior Citizens Act of 2003
- PD No. 881, or The Household Hazardous Act
- EO No. 51, or The Milk Code of the Philippines
- RA 10354, or The Responsible Parenthood and Reproductive Health
Bill of 2012
- PD 856, or The Code of Sanitation of the Philippines
To guarantee the safety, quality, purity, efficacy of health products in order to protect and promote the right to health of the general public.
To be an internationally recognized center of excellence in health product regulation by 2026.
In order to protect and promote the right to health of the Filipino people and to establish and maintain an effective health products regulatory system responsive to the country’s health needs and problems, the FDA shall perform the following functions as provided by existing laws:
- To establish safety or efficacy standards and quality measures for foods, drugs and devices and cosmetics and other health product;
- To undertake appropriate health manpower development and research, responsive to the country’s health needs and problems;
- To assume primary jurisdiction in the collection of samples of health products;
- To analyze and inspect health products;
- To establish analytical data to serve as basis for the preparation of health products standards, and to recommend standards of identity, purity, safety, efficacy, quality and fill of container;
- To issue certificates of compliance with technical requirements to serve as basis for the issuance of appropriate authorization and spot-check for compliance with regulations regarding operation of manufacturers, importers, exporters, distributors, wholesalers, drug outlets, and other establishments and facilities of health products, as determined by the FDA;
- To conduct appropriate tests on all applicable health products prior to the issuance of appropriate authorizations to ensure safety, efficacy, purity, and quality;
- To require all manufacturers, traders, distributors, importers, exporters, wholesalers, retailers, consumers, and non-consumer users of health products to report to the FDA any incident that reasonably indicates that said product has caused or contributed to the death, serious illness or serious injury to a consumer, a patient, or any person;
- To issue cease and desist orders motu propio or upon verified complaint for health produts, whether or not registered with the FDA Provided, That for registered health products, the cease and desist order is valid for thirty (30) days and may be extended for sixty (60) days only after due process has been observed;
- To order the ban, recall, and/or withdrawal of any health product, after due process, found to have caused the death, serious illness or serious injury to a consumer or patient, or is found to be imminently injurious, unsafe, dangerous, or grossly deceptive, and to require all concerned to implement the risk management plan which is a requirement for the issuance of the appropriate authorization;
- To strengthen the post market surveillance system in monitoring health products as defined in this Act and incidents of adverse events involving such products;
- To develop and issue standards and appropriate authorizations that would cover establishments, facilities and health products;
- To conduct, supervise, monitor and audit research studies on health and safety issues of health products undertaken by entities duly approved by the FDA;
- To prescribe standards, guidelines, and regulations with respect to information, advertisements and other marketing instruments and promotion, sponsorship, and other marketing activities about the health products;
- To maintain bonded warehouses and/or establish the same, whenever necessary or appropriate, as determined by the director-general for confiscated goods in strategic areas of the country especially at major ports of entry; and
- To exercise such other powers and perform such other functions as may be necessary to carry out its duties and responsibilities.
- To protect and promote the right to health of the Filipino people by ensuring the safety, efficacy, quality, and purity of foods, drugs, devices, and cosmetics, and
- To establish and maintain an effective health products regulatory system responsive to the country’s health needs and problems.
Ensure the safety, efficacy, quality and purity of health products by fostering integrity, transparency and excellence; developing and maintaining evidence-based standards and policies, in a healthy and safe work environment.
Bawat Kawani ng FDA, Lingkod Bayani"
EXCELLENCE - Pagiging Mahusay refers to our Highest degree of professionalism and superior work standard in discharging one's duty
LEADERSHIP - Mahusay na Pamumuno refers to the management thinking that all employees in all levels of public service who, at their best, care and do something about the challenges faced by the FDA
INTEGRITY - Pagiging Matapat refers to our consistent adherence to strong and ethical principles, wether alone or in public
PATRIOTISM - Pagiging Makabayan refers to our love for our country and fellowmen
SPIRITUALITY - Pagiging Maka-Diyos refers to our belief, love and faith for a Higher Being
 |
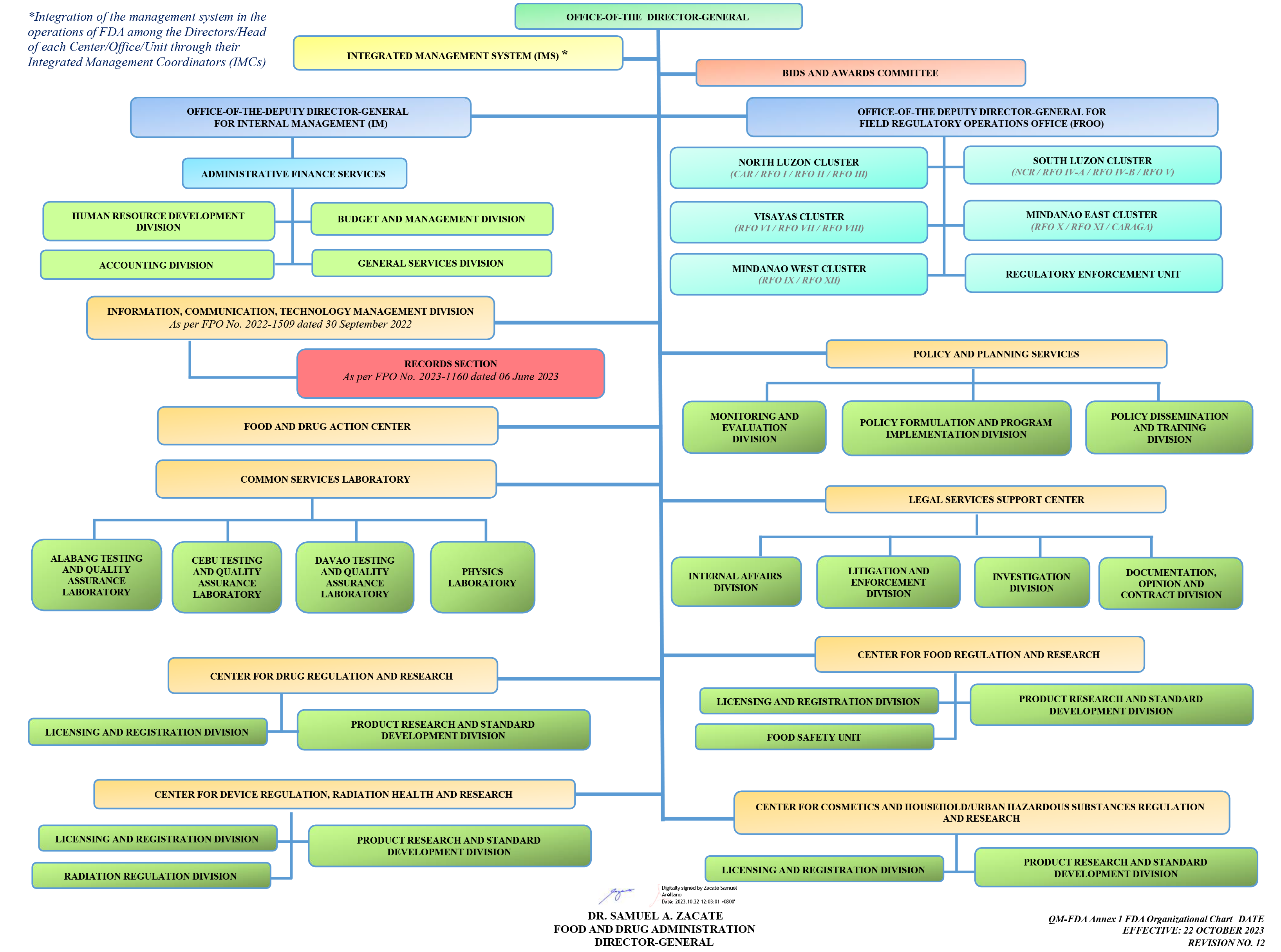
Dr. Samuel A. Zacate, FICS FPSVI DPCOM CSPSH, Director General |
Office of the Director General |
 |
Atty. Ronald R. De Veyra, MBA, CESO II,
Deputy Director General for Internal Management |
Office of the Deputy Director General for Internal Management |
 |
Dr. Oscar G. Gutierrez, Jr., MPA, PCVPH,
Deputy Director General for Field Regulatory Operations Office |
Office of the Deputy Director General for Field Regulatory Operations Office |
 |
Atty. John Benedict G. Aguzar,
Director III |
Administrative and Finance Service |
 |
Engr. Ana Trinidad F. Rivera,
Director IV |
Center for Cosmetics and Household Urban Hazardous Substances Regulation and Research |
 |
Jesusa Joyce N. Cirunay,
Director IV |
Center for Drug Regulation and Research |
 |
Maria Cecilia C. Matienzo,
Director IV |
Center for Device Regulation, Radiation and Health Research |
 |
Pilar Marilyn M. Pagayunan,
Director IV |
Center for Food Regulation and Research |
 |
Jocelyn E. Balderrama, RPh., MBA,
Director III |
Common Services Laboratory |
 |
Atty. Emilio L. Polig, Jr.,
Director III |
Legal Services Support Center |
 |
Estrellita B. Pastolero, MGM,
Officer-In-Charge |
Food and Drug Action Center |
 |
Irene Florentino-Fariñas, RPh., MD, MNSA,
Director III |
Policy and Planning Service |
 |
Gomel C. Gabuna, LlB, MDM, CESE,
Director II |
North Luzon Cluster, Field Regulatory Operations Officer |
 |
Arnold G. Alindada, Dr. HCM, MPH, RMT,
Director II |
South Luzon Cluster, Field Regulatory Operations Office |
 |
Rica Reina C. Aumentado,
Director II, Officer-In-Charge |
Visayas Cluster, Field Regulatory Operations Office |
 |
Deborah S. Legaspi,
Director II |
Mindanao East Cluster, Field Regulatory Operations Office |
 |
Sharon Rose P. Garcia,
Director II, Officer-In-Charge |
Mindanao West Cluster, Field Regulatory Operations Office |


















