FDA Advisory No. 2020-2110 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Terbinafine (Noali®) 10mg/g (1%) Cream 2. 1-Ostealgia Plaster Far-Infrared […]
FDA Advisory No. 2020-2109-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. Topfond® Gentamycin Sulfate Injection 2. Epinephrine Hydrochloride Injection 1ml:1mg 3. Furosemide […]
FDA Advisory No. 2020-2109 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Topfond® Gentamycin Sulfate Injection 2. Epinephrine Hydrochloride Injection 1ml:1mg […]
FDA Advisory No. 2020-2108-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. OTC Houtongling Keli 2. OTC Kesutingtangjiang 100ml 3. Cefradine For Injection […]
FDA Advisory No. 2020-2108 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Houtongling Keli 2. OTC Kesutingtangjiang 100ml 3. Cefradine […]
FDA Advisory No. 2020-2107-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. Compound Aminophenazone and Barbital Injection 2mL 2. Calcium Gluconate Injection 10mg:1g […]
FDA Advisory No. 2020-2107 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Compound Aminophenazone and Barbital Injection 2mL 2. Calcium Gluconate […]
FDA Advisory No. 2020-2106-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. Povidone Betadine 15ml 2. Povidone Betadine 60ml 3. Methyl Salicylate (Oil […]
FDA Advisory No. 2020-2106 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Povidone Betadine 15ml 2. Povidone Betadine 60ml 3. Methyl […]
FDA Advisory No. 2020-2105-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:
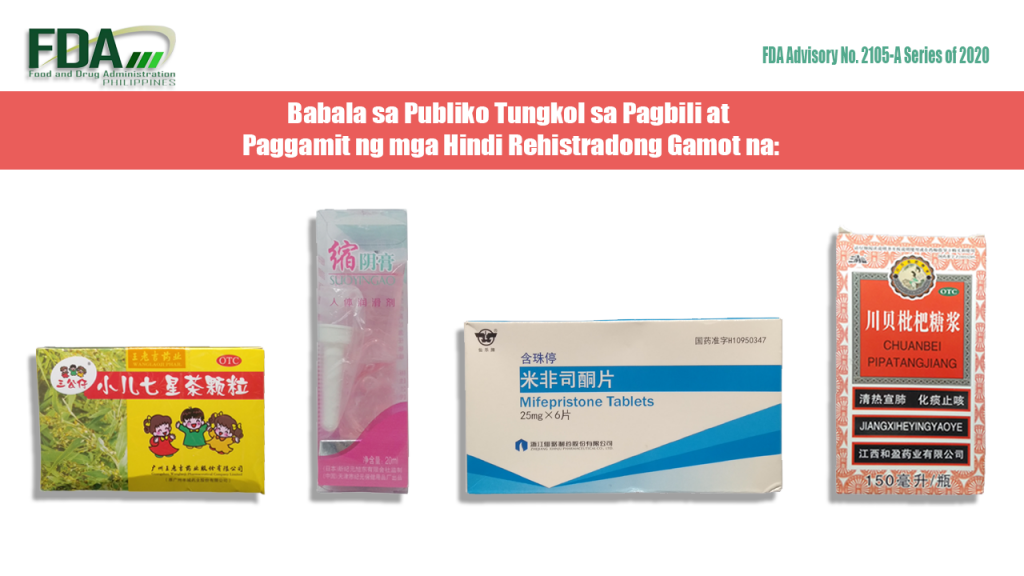
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. OTC Xiao’er Qixingcha Keli 2. Suyingao 20ml 3. Mifepristone Tablets 25mg […]



