FDA Advisory No.2023-1938 || Public Health Warning Against the Purchase and Use of the Following Unnotified Medical Device Products:

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the following unnotified medical device products: 1. 1. “RX DR. CARE™ […]
FDA Advisory No.2023-1937 || Public Health Warning Against the Purchase and Use of the Unregistered Medical Device “MEDX ALL-SILICONE DUODENAL TUBE FR18”
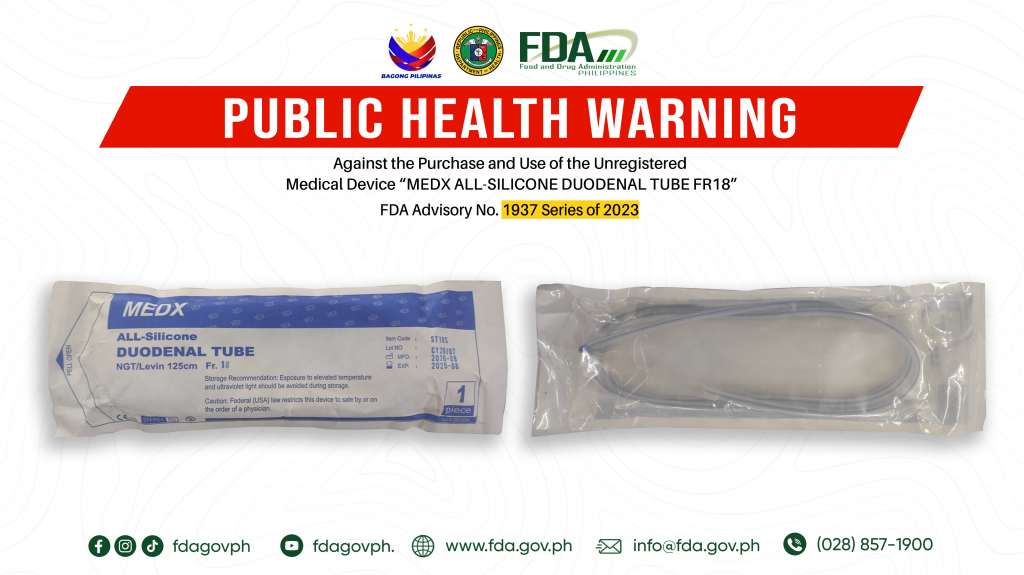
The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the unregistered medical device product: 1. MEDX ALL-SILICONE DUODENAL TUBE FR18 […]
FDA Advisory No.2023-1936 || Public Health Warning Against the Purchase and Use of the Unnotified Medical Device Product “WELLCARE KINESIOLOGY MUSCLE TAPE”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the unnotified medical device product: 1. WELLCARE KINESIOLOGY MUSCLE TAPE The […]
FDA Advisory No.2023-1935 || Public Health Warning Against the Purchase and Use of the Unregistered Health-Related Device Product “AGUAWATER ALKALINE WATER PURIFIER”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the unregistered health-related device product: 1. AGUAWATER ALKALINE WATER PURIFIER The […]
FDA Advisory No.2023-1934 || Public Health Warning Against the Purchase and Use of the Unnotified Medical Device Product “RX DR. CARE EXAMINATION GLOVES VINYL GLOVES POWDER-FREE”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the unnotified medical device product: 1. RX DR. CARE EXAMINATION GLOVES […]
FDA Advisory No.2023-1928 || Public Health Warning Against the Purchase and Use of the Unnotified Medical Device Product “PEAK FLOW METER”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the unnotified medical device product: 1. PEAK FLOW METER The FDA […]
FDA Advisory No.2023-1927 || Public Health Warning Against the Purchase and Use of the Following Unregistered MEDICAL DEPOT Medical Device Products:

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the unregistered medical device products: 1. “OXYGEN MASK WITH TUBING (ADULT […]
FDA Advisory No.2023-1926 || Public Health Warning Against the Purchase and Use of the Misbranded Medical Device Product “Pulse Oximeter (In Foreign Characters)”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the misbranded medical device product: 1. Pulse Oximeter (In Foreign Characters) […]
FDA Advisory No.2023-1892 || Public Health Warning Against the Purchase and Use of the Banned Medical Device “MAXWELL POWDERED SMOOTH SURFACE LATEX EXAMINATION GLOVES”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the banned medical device product: 1. MAXWELL POWDERED SMOOTH SURFACE LATEX […]
FDA Advisory No.2023-1891 || Public Health Warning Against the Purchase and Use of the Unnotified Medical Device Product “ELASTIC BANDAGE HONEYCOMB”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the unnotified medical device product: 1. ELASTIC BANDAGE HONEYCOMB The FDA […]



