FDA Advisory No.2024-0519 || Public Health Warning Against the Purchase and Use of the Unregistered Medical Device Product “ETHICON® PROLENE™ POLYPROPYLENE ETHALLOY™ NEEDLE ALLOY”

The Food and Drug Administration (FDA) warns all healthcare professionals and the general public NOT TO PURCHASE AND USE the unregistered medical device product: 1. ETHICON® PROLENE™ POLYPROPYLENE ETHALLOY™ NEEDLE […]
FDA Advisory No.2024-0501-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at Paggamit ng mga sumusunod na hindi rehistradong gamot: 1. Pharm® Tuobaokewo Fenspian – Cefixime Dispersible Tablets [as […]
FDA Advisory No.2024-0502-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot na “Minoxidil Nexxus Hair Regrowth Treatment 6% Minoxidil 60 mL”
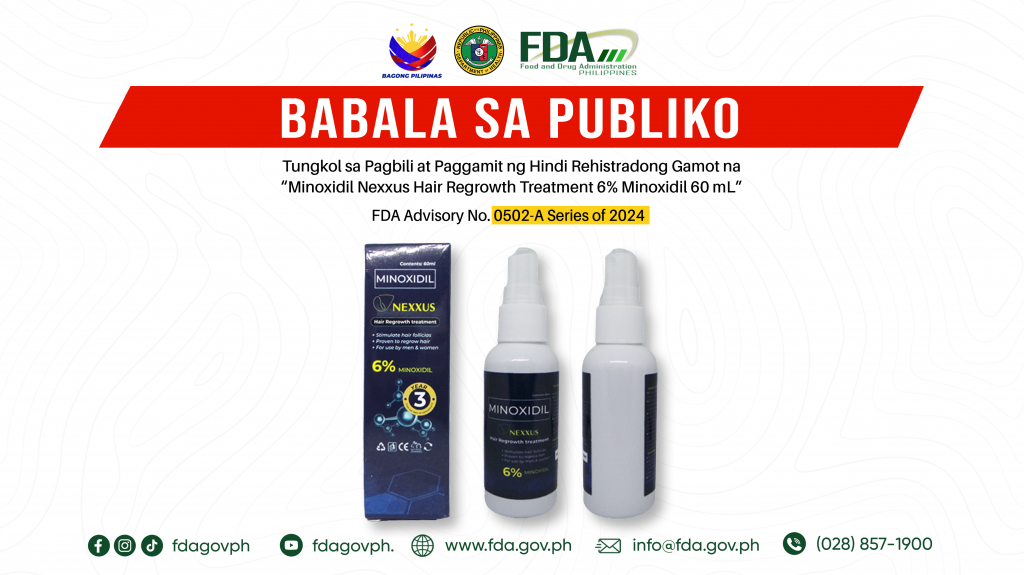
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot na: 1. Minoxidil Nexxus Hair Regrowth Treatment 6% Minoxidil 60 mL Napatunayan […]
FDA Advisory No.2024-0502 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “Minoxidil Nexxus Hair Regrowth Treatment 6% Minoxidil 60 mL”

The Food and Drug Administration (FDA) advises the public against the purchase and use of the unregistered drug product: 1. Minoxidil Nexxus Hair Regrowth Treatment 6% Minoxidil 60 mL FDA […]
FDA Advisory No.2024-0501 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Pharm® Tuobaokewo Fenspian – Cefixime Dispersible Tablet [as reflected […]
FDA Advisory No.2024-0500-A || Babala sa Publiko tungkol sa Paggamit ng Beripikadong Pekeng Gamot na “Valium® (Diazepam) 10 mg/2 mL Solution for Injection I.M./I.V. 5 ampoules of 2 mL”

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng beripikadong pekeng gamot: 1. Valium® (Diazepam) 10 mg/2 mL Solution for Injection I.M./I.V. 5 ampoules […]
FDA Advisory No.2024-0500 || Public Health Warning Against the Purchase and Use of the Counterfeit Drug Product “Valium® (Diazepam) 10 mg/2 mL Solution for Injection I.M./I.V. 5 ampoules of 2 mL”

The Food and Drug Administration (FDA) advises the public against the purchase and use of the counterfeit version of the following product: 1. Valium® (Diazepam) 10 mg/2 mL Solution for […]
FDA Advisory No.2024-0429 || Clarification on the Requirements for Senior Citizens to Avail 20% Discount on Medicines

This is to reiterate the coverage and requirements of the 20% discount on purchase of medicines for Senior Citizens. Section IV.2 of Administrative Order (AO) No. 2012-0007-A entitled, “Amendment to […]
FDA Advisory No.2024-0337-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. Huons Hycomin Inj. (Hydroxocobalamin) 5 mg/ 2 mL […]
FDA Advisory No.2024-0337 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Huons Hycomin Inj. (Hydroxocobalamin) 5 mg/ 2 mL x […]



