FDA Advisory No.2024-0337-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. Huons Hycomin Inj. (Hydroxocobalamin) 5 mg/ 2 mL […]
FDA Advisory No.2024-0337 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Huons Hycomin Inj. (Hydroxocobalamin) 5 mg/ 2 mL x […]
FDA Advisory No.2024-0336-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:
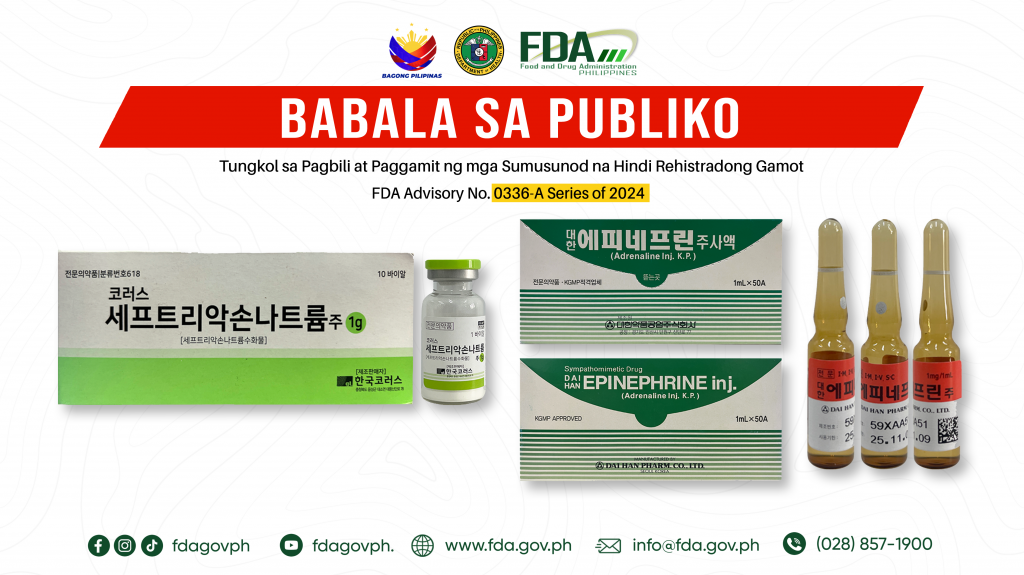
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. [Label in foreign language] 1g Vial 2. DAI […]
FDA Advisory No.2024-0336 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. [Label in foreign language] 1g Vial 2. DAI HAN […]
FDA Advisory No.2024-0335-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot na “Miao Relieves Headache Qu Feng You 10 mL [As reflected in the secondary packaging] Miao Qu Feng You Relieves Headache 10 mL [As reflected in the primary packaging].”

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot na: 1. Miao Relieves Headache Qu Feng You 10 mL [As reflected […]
FDA Advisory No.2024-0335 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Product:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug product: 1. Miao Relieves Headache Qu Feng You 10 mL [As […]
FDA Advisory No.2024-0334-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at Paggamit ng mga sumusunod na hindi rehistradong gamot: 1. Himalaya® Koflet® Cough Syrup 100 mL 2. ZANDU […]
FDA Advisory No.2024-0334 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Himalaya® Koflet® Cough Syrup 100 mL 2. ZANDU Nityam […]
FDA Advisory No.2024-0333-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at Paggamit ng mga sumusunod na hindi rehistradong gamot: 1. SHIN POONG Antispasmodic Eukinetic Agent Tiram Injection Tiropramide […]
FDA Advisory No.2024-0333 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. SHIN POONG Antispasmodic Eukinetic Agent Tiram Injection Tiropramide HCL […]



