FDA Advisory No. 2020-1614-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:
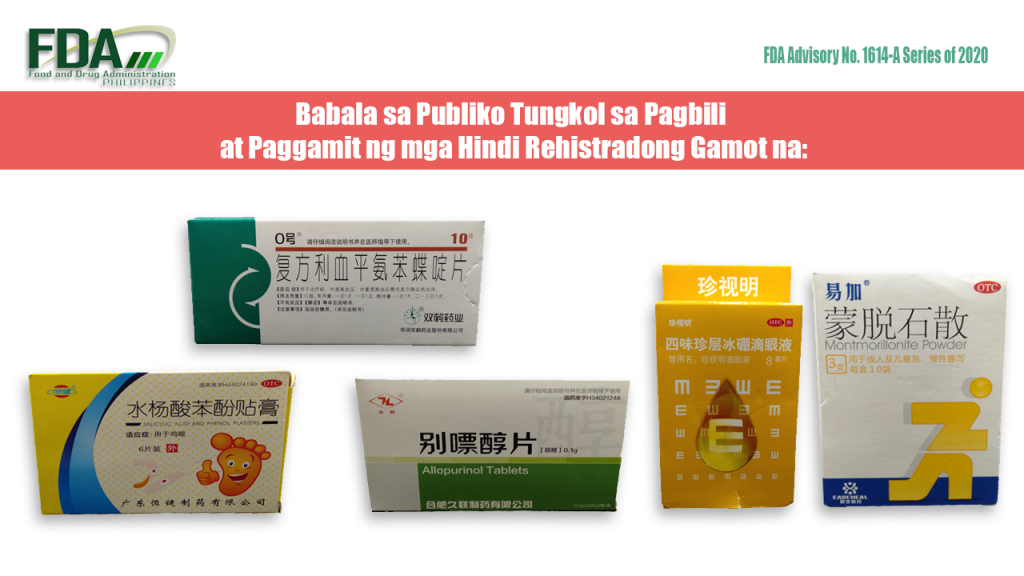
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. OTC Salicylic Acid and Phenol Plasters 2. TL® Allopurinol […]
FDA Advisory No. 2020-1614 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Salicylic Acid and Phenol Plasters 2. TL® Allopurinol 0.1g […]
FDA Advisory No. 2020-1613-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. Ruhe Sanjie Tablets 2. Vitamin E (as reflected in […]
FDA Advisory No. 2020-1613 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Ruhe Sanjie Tablets 2. Vitamin E (as reflected in […]
FDA Advisory No. 2020-1612-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. OTC Naoxinshu Koufuye 2. Apollo® Hericium Erinaceus Super-Quality Healthy […]
FDA Advisory No. 2020-1612 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Naoxinshu Koufuye 2. Apollo® Hericium Erinaceus Super-Quality Healthy […]
FDA Advisory No. 2020-1611-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. Ejiao 10g 2. OTC Jing Wan Hong Ruan Gao […]
FDA Advisory No. 2020-1611|| Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Ejiao 10g 2. OTC Jing Wan Hong Ruan Gao […]
FDA Advisory No. 2020-1610-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. OTC Paracetamol, Caffeine and Aspirin Powder 2. OTC Yunnan […]
FDA Advisory No. 2020-1610 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Paracetamol, Caffeine and Aspirin Powder 2. OTC Yunnan […]



