Announcement || Reminder on the Submission of Financial Reports in the FDA Online Disclosure Reporting System

Pursuant to Section 35(b) of RA No. 11223 on Ethics in Public Health Policy and Practice, all FDA-licensed manufacturers, traders, repackers, distributor-importers, and distributor- wholesalers of drug, medical devices, and […]
Draft for Comments || Guidelines on the Importation and Exportation Notification for Pharmaceutical Products and Raw Materials

All comments may be sent to [email protected]. The deadline for submission of comments is on 20 May 2024. Please see attached files for your reference. Attachments Draft for Comments (2 MB)TEMPLATE-COMMENT-FORM […]
NP-SVP(A)-2024-0087-RFOXI || Procurement of Services for Cleaning and Installation of Dismantled Air-conditioning Units in the New Office Space of FDA Regional Field Office XI

Project Descriptions Bid Bulletins/Issuances/ Announcements/Remarks Status Procurement of Services for Cleaning and Installation of Dismantled Air-conditioning Units in the New Office Space of FDA Regional Field Office XI […]
FDA Advisory No.2024-0669 || Public Health Warning Against the Purchase and Use of Unnotified Cosmetic “DEOPROCE SKIN RESCUE LIGHTWEIGHT SUNSCREEN SPF50 PA+++ 06”

The Food and Drug Administration (FDA) warns the public NOT TO PURCHASE AND USE unnotified cosmetic product, DEOPROCE SKIN RESCUE LIGHTWEIGHT SUNSCREEN SPF50 PA+++ 06. The abovementioned product was verified […]
FDA Advisory No.2024-0668 || Public Health Warning Against the Purchase and Use of Unnotified Cosmetic “SANTA MARIA VIRGIN COCONUT OIL ALOE VERA”
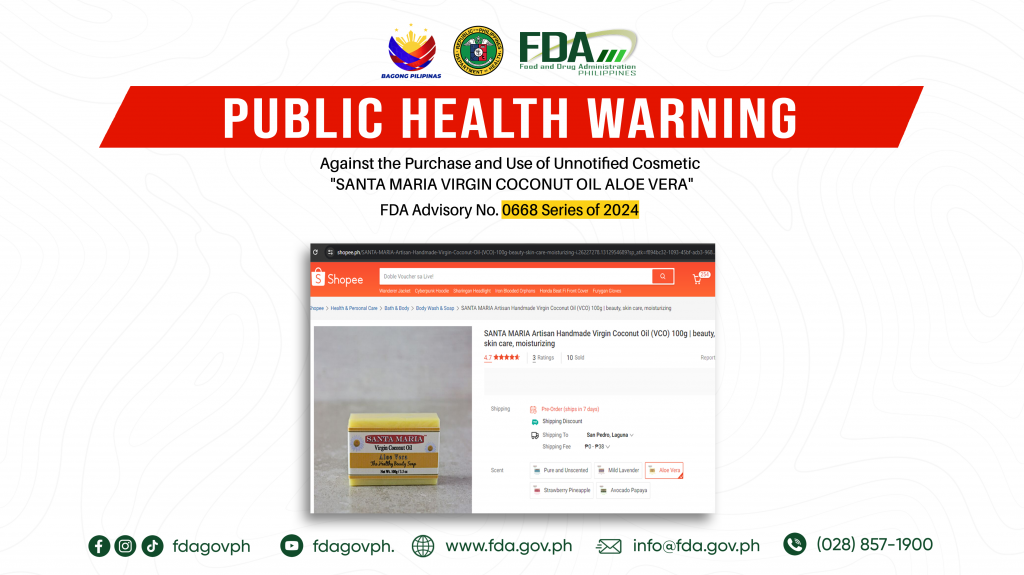
The Food and Drug Administration (FDA) warns the public NOT TO PURCHASE AND USE unnotified cosmetic product, SANTA MARIA VIRGIN COCONUT OIL ALOE VERA. The abovementioned product was verified by […]
FDA Advisory No.2024-0667 || Public Health Warning Against the Purchase and Use of Unnotified Cosmetic “SOUG WELL HAIR REPAIR AND MAINTENANCE HAIR MASK”

The Food and Drug Administration (FDA) warns the public NOT TO PURCHASE AND USE unnotified cosmetic product, SOUG WELL HAIR REPAIR AND MAINTENANCE HAIR MASK. The abovementioned product was verified […]
FDA Advisory No.2024-0666 || Public Health Warning Against the Purchase and Use of Unnotified Cosmetic “SY GLOW DEO PROTECT TONER”

The Food and Drug Administration (FDA) warns the public NOT TO PURCHASE AND USE unnotified cosmetic product, SY GLOW DEO PROTECT TONER. The abovementioned product was verified by FDA through […]
FDA Advisory No.2024-0665 || Public Health Warning Against the Purchase and Use of Unnotified Cosmetic “SY GLOW WHITENING BEAUTY DEO INSTANT WHITE PROTECT CREAM”

The Food and Drug Administration (FDA) warns the public NOT TO PURCHASE AND USE unnotified cosmetic product, SY GLOW WHITENING BEAUTY DEO INSTANT WHITE PROTECT CREAM. The abovementioned product was […]
FDA Advisory No.2024-0664 || Public Health Warning Against the Purchase and Use of Unnotified Cosmetic “SANTA MARIA VIRGIN COCONUT OIL – MILD”
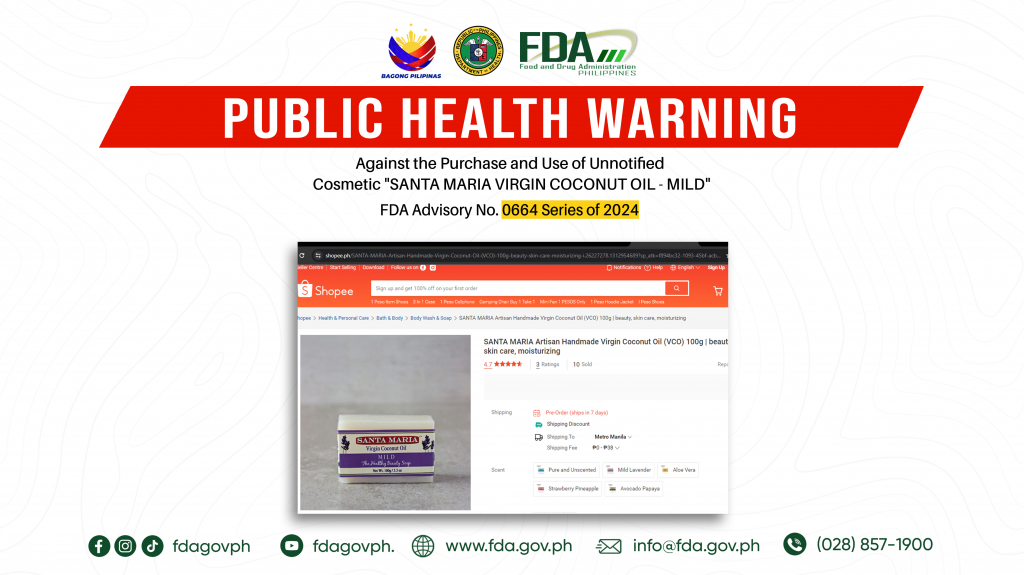
The Food and Drug Administration (FDA) warns the public NOT TO PURCHASE AND USE unnotified cosmetic product, SANTA MARIA VIRGIN COCONUT OIL – MILD. The abovementioned product was verified by […]
FDA Advisory No.2024-0663 || Public Health Warning Against the Purchase and Use of Unnotified Cosmetic “SUNISA ORANGE EXFOLIATING WHITENING GEL”

The Food and Drug Administration (FDA) warns the public NOT TO PURCHASE AND USE unnotified cosmetic product, SUNISA ORANGE EXFOLIATING WHITENING GEL. The abovementioned product was verified by FDA through […]



