FDA Advisory No.2021-0362 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Levofloxacin Hydrochloride Tablets 0.2g 2. OTC TAIJI Huoxiang Zhenqi […]
FDA Advisory No.2021-0361-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. OTC Dieda Wanhua You 2. OTC Ambroxol Hydrochloride Tablets – Yansuan […]
FDA Advisory No.2021-0361 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Dieda Wanhua You 2. OTC Ambroxol Hydrochloride Tablets […]
FDA Advisory No.2021-0360-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Pekeng Bersyon ng Purified Rabies Vaccine (Vero Cell) Speeda 2.5 I.U. and 0.5 mL of Solvent Freeze-Dried Powder for Injection

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga pekeng bersyon ng Purified Rabies Vaccine (Vero Cell) Speeda 2.5 I.U. and 0.5 mL […]
FDA Advisory No.2021-0360 || Public Health Warning Against the Purchase and Use of the Counterfeit Purified Rabies Vaccine (Vero Cell) Speeda 2.5 I.U. and 0.5 mL of Solvent Freeze-Dried Powder for Injection
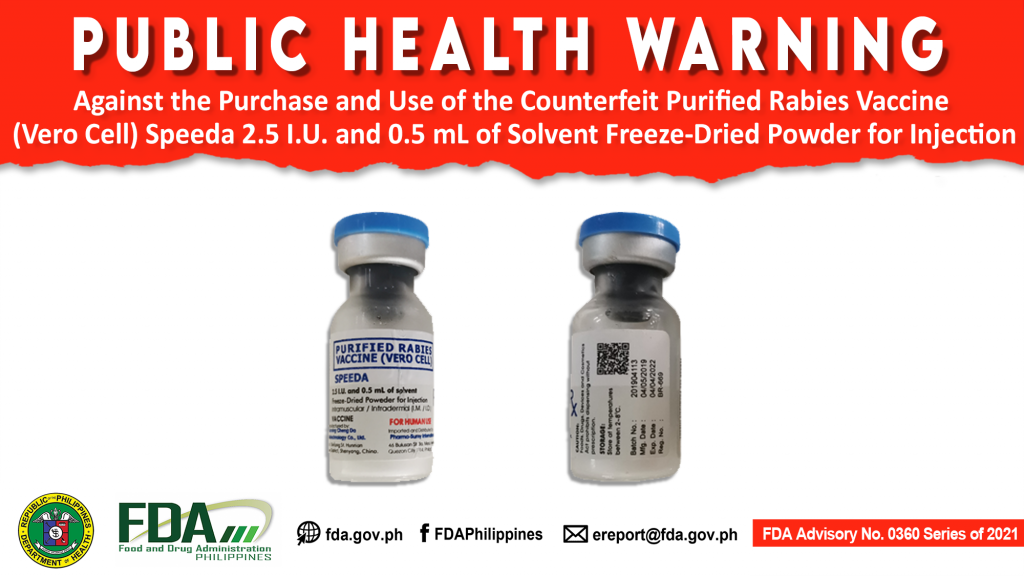
The Food and Drug Administration (FDA) advises the public against the purchase and use of the counterfeit Purified Rabies Vaccine (Vero Cell) Speeda 2.5 I.U. and 0.5 mL of Solvent […]
FDA Advisory No.2021-0351 || Product Recall of Specific Lots of Neostigmine Methylsulfate 0.50 mg/mL Solution for Injection (IM/IV/SC) (Prostigmin)

Drug distributors and drug wholesalers are hereby advised by the Food and Drug Administration (FDA) regarding the recall of the affected lots of the subject product. The details of the […]
FDA Advisory No.2021-0352 || Product Recall of Specific Batches of Three (3) Dosage Forms of Deproteinized Calf Blood Extract (Solcoseryl)
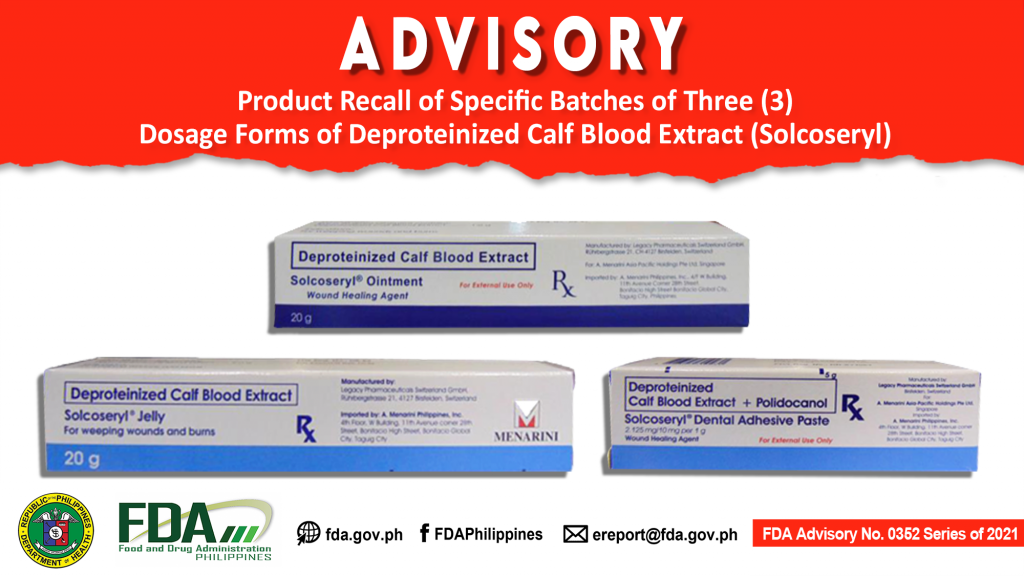
Drug distributors and drug wholesalers are hereby advised by the Food and Drug Administration (FDA) regarding the recall of the affected batches of the subject products. The details of the […]
FDA Advisory No.2021-0340 || Public Health Warning Against the Purchase and Use of the following Unauthorized Cosmetic Products:
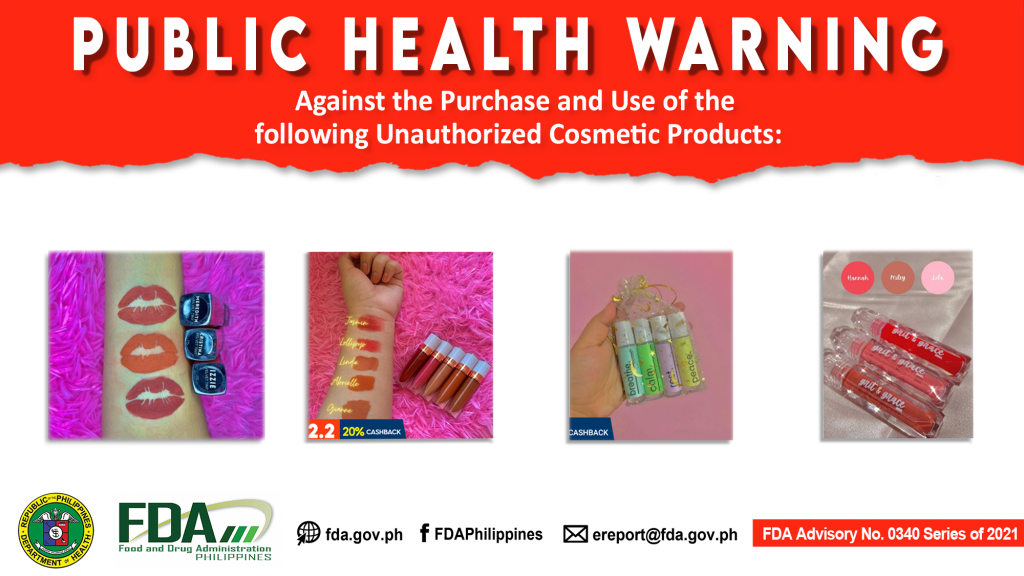
The Food and Drug Administration (FDA) warns the public from purchasing and using the following unauthorized cosmetic products. 1. GRIT & GRACE MNL VELVET TINTS 2. GRIT & GRACE MNL […]
FDA Advisory No.2021-0339 || Public Health Warning Against the Purchase and Use of Unauthorized Cosmetic HYDRO GEL WATER BASED HAIR STYLING GEL

The Food and Drug Administration (FDA) warns the public from purchasing and using the unauthorized cosmetic product, HYDRO GEL WATER BASED HAIR STYLING GEL . The abovementioned product was verified by […]
FDA Advisory No.2021-0338 || Public Health Warning Against the Purchase and Use of the Non-Compliant Cosmetic Product ” BENCH/ BRATSPLASH INSTANT HAND SANITIZER GEL WITH MOISTURIZING ALOE KIWI FROST “

The Food and Drug Administration (FDA) warns the general public from purchasing and using the non-compliant cosmetic product, ” BENCH/ BRATSPLASH INSTANT HAND SANITIZER GEL WITH MOISTURIZING ALOE KIWI FROST “. […]



