FDA Advisory No.2024-0641-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot “Lido-Jel Topical Anesthetic Gel Strawberry Flavor 30GMS”

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot: 1. Lido-Jel Topical Anesthetic Gel Strawberry Flavor 30GMS Napatunayan sa pamamagitan ng […]
FDA Advisory No.2024-0641 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “Lido-Jel Topical Anesthetic Gel Strawberry Flavor 30GMS”

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug product: 1. Lido-Jel Topical Anesthetic Gel Strawberry Flavor 30GMS FDA Post-Marketing […]
FDA Advisory No.2024-0637-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. Ropivacaine HCl Injection, USP 0.5% 150 mg/ 30 […]
FDA Advisory No.2024-0637 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Ropivacaine HCl Injection, USP 0.5% 150 mg/ 30 mL […]
FDA Advisory No.2024-0639 || Clarification on the Requirements for Change of Qualified Person Initially Registered in a Licensed Drug Establishment

Section V.B.3.C of FDA Circular No. 2020-030, entitled “Guideline for the Use of the FDA eServices Portal System for License to Operate (LTO) Application of Drug Distributors, Drug Traders, Drugstores, […]
FDA Advisory No.2024-0576-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. OTC Clindamycin Phosphate Gel 2. OTC Z36021451 [label […]
FDA Advisory No.2024-0576 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Clindamycin Phosphate Gel 2. OTC Z36021451 [Label in […]
FDA Advisory No.2024-0575-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:
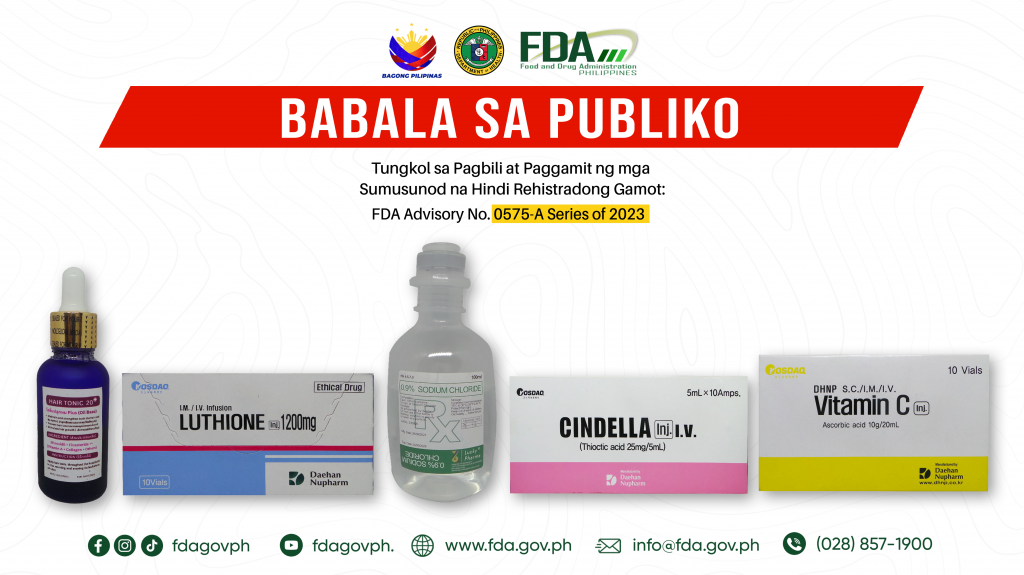
The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Hair Tonic 20 Minoxidil + Finasteride ++ 2. KOSDAQ […]
FDA Advisory No.2024-0575 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Hair Tonic 20 Minoxidil + Finasteride ++ (Vitamin A […]
FDA Advisory No.2024-0574-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. OTC Ibuprofen Granules 2. Zang Jikang® (as translated) […]



