FDA Advisory No.2024-0742-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot “Recombinant Human Erythropoietin Injection IP 4000 IU (Renocrit ™ 4000) Single dose pre-filled syringe of 4000 IU/ 0.4 mL”
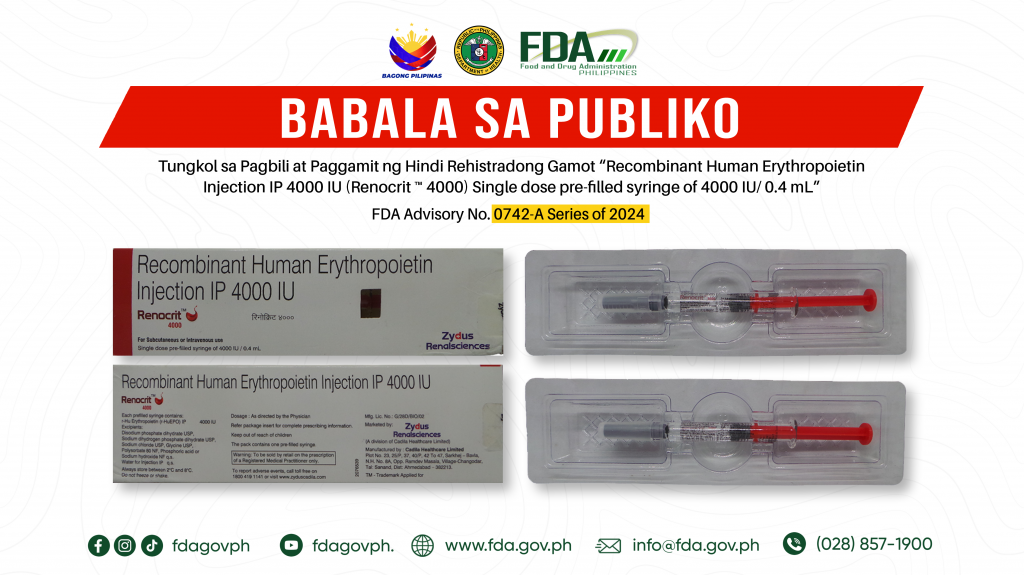
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot: 1. Recombinant Human Erythropoietin Injection IP 4000 IU (Renocrit ™ 4000) Single […]
FDA Advisory No.2024-0742 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “Recombinant Human Erythropoietin Injection IP 4000 IU (Renocrit ™ 4000) Single dose pre-filled syringe of 4000 IU/ 0.4 mL”
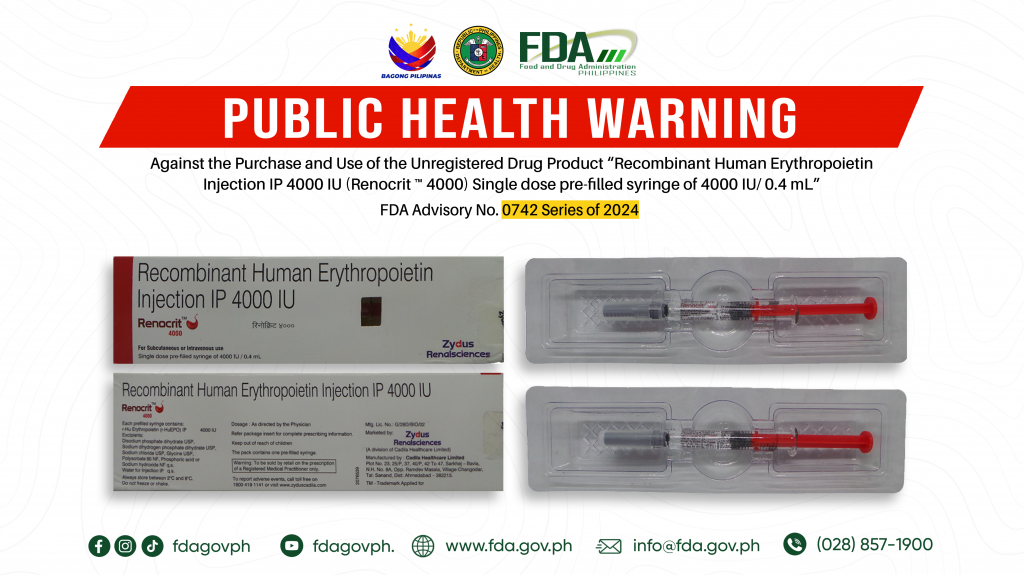
The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug product: 1. Recombinant Human Erythropoietin Injection IP 4000 IU (Renocrit ™ […]
FDA Advisory No.2024-0741 || Product Recall of All Available Batches of Methotrexate 100 mg/mL Solution for Injection (IM/IV) [Trexasaph]

All healthcare professionals and the general public are hereby warned by the Food and Drug Administration (FDA) that the affected batches of the subject product are being recalled from the […]
FDA Advisory No.2024-0740 || Product Recall of Specific Lot of Enalapril Maleate 10 mg Tablet [Scheepril]

All healthcare professionals and the general public are hereby warned by the Food and Drug Administration (FDA) that the affected lot of the subject product is being recalled from the […]
FDA Advisory No.2024-0640-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. OTC LN Bromohexine Hydrochloride Tablet 2. OTC Shumi […]
FDA Advisory No.2024-0640 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Shumi Jiaonang [as reflected in package insert] 2. […]
FDA Advisory No.2024-0577-A || Babala sa Publiko tungkol sa Paggamit ng Beripikadong Pekeng Gamot na “Human Tetanus Immunoglobulin (Tetagam® P) 1 ml (250 IU) Solution for Injection”
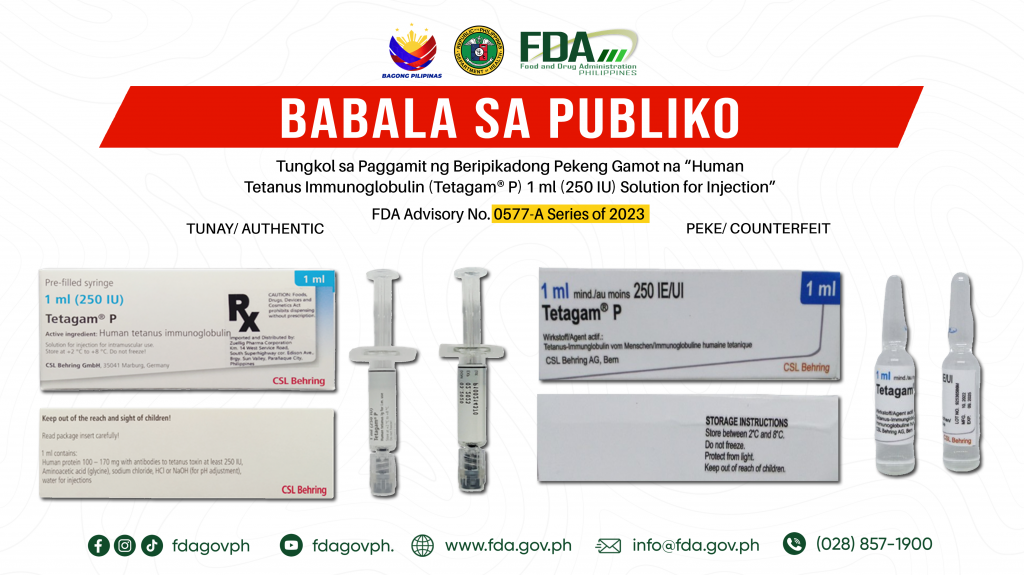
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng beripikadong pekeng gamot: 1. Human Tetanus Immunoglobulin (Tetagam® P) 1 ml (250 IU) Solution for […]
FDA Advisory No.2024-0577 || Public Health Warning Against the Purchase and Use of the Verified Counterfeit Drug Product “Human Tetanus Immunoglobulin (Tetagam® P) 1 ml (250 IU) Solution for Injection”
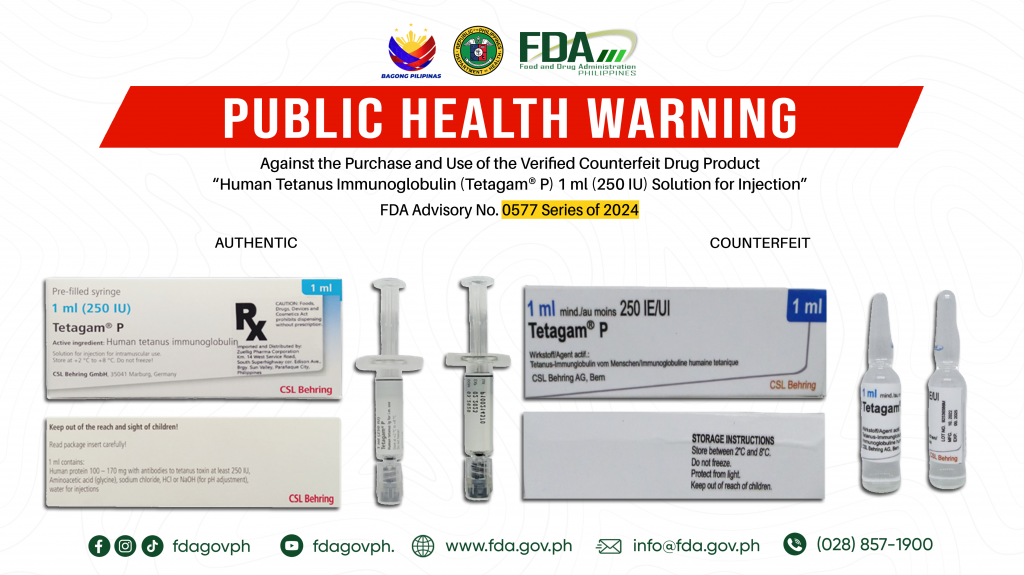
The Food and Drug Administration (FDA) advises the public against the purchase and use of the counterfeit version of the following product: 1. Human Tetanus Immunoglobulin (Tetagam® P) 1 ml […]
FDA Advisory No.2024-0653-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot “Bodymate 70% Isopropyl Alcohol Antiseptic Sanitizer 500 mL”

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot: 1. Bodymate 70% Isopropyl Alcohol Antiseptic Sanitizer 500 mL” Napatunayan sa pamamagitan […]
FDA Advisory No.2024-0653 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “Bodymate 70% Isopropyl Alcohol Antiseptic Sanitizer 500 mL”

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug product: 1. Bodymate 70% Isopropyl Alcohol Antiseptic Sanitizer 500 mL FDA […]



