The Food and Drug Administration (FDA) notifies the public on the WHO Medical Product Alert on four (4) substandard (contaminated) pediatric drug products which were detected in the African region in September 2022:
| PRODUCT NAME |
Promethazine Oral Solution BP |
Pheniramine Maleate, Ammonium Chloride, Menthol
[Kofexmalin Baby Cough Syrup] |
| STATED MANUFACTURER |
Maiden Pharmaceuticals Ltd. |
Maiden Pharmaceuticals Ltd. |
|
LOT |
ML21-202 |
ML21-199 |
|
EXP. DATE |
Nov-24 |
Nov-24 |
|
PACKAGING LANGUAGE |
English |
English |
|
AVAILABLE PHOTO |

|
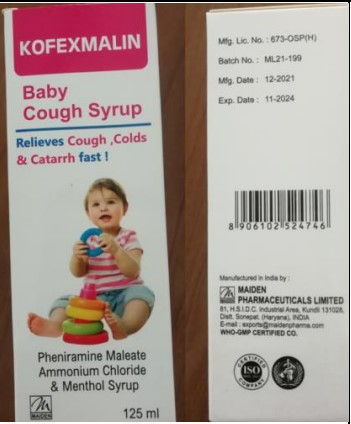 |
Figure 1. Promethazine Oral Solution BP and Pheniramine Maleate, Ammonium Chloride, Menthol [Kofexmalin Baby Cough Syrup]detected in the African region
| PRODUCT NAME |
Chlorphenamine Maleate, Phenylephrine HBr, Dextromethorphan [MaKOFF Baby Cough Syrup] |
Paracetamol Phenylephrine HCl, Chlorphenamine Maleate [MaGrip n Cold Syrup] |
| STATED MANUFACTURER |
Maiden Pharmaceuticals Ltd. |
Maiden Pharmaceuticals Ltd. |
|
LOT |
ML21-203 |
ML21-198 |
|
EXP. DATE |
Nov-24 |
Nov-24 |
|
PACKAGING LANGUAGE |
English |
English |
|
AVAILABLE PHOTO |
|
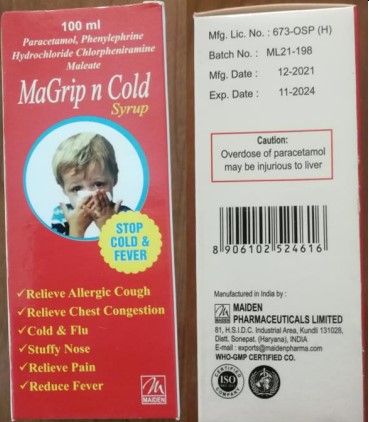 |
Figure 2. Chlorphenamine Maleate, Phenylephrine HBr, Dextromethorphan [MaKOFF Baby Cough Syrup] and Paracetamol Phenylephrine HCl, Chlorphenamine Maleate [MaGrip n Cold Syrup] detected in the African region
The FDA strongly advises the public to be vigilant on the circulation of these substandard drug products since its contaminants, Diethylene Glycol and Ethylene Glycol, are toxic to humans when consumed and may result to abdominal pain, vomiting, diarrhea, inability to pass urine, headache, altered mental state and acute kidney injury which may lead to death. A substandard drug product are products that fail to meet either their quality standards or specifications. To date, the stated manufacturer has not provided guarantees to WHO on the safety and quality of these products. All batches of these products should be considered unsafe until they can be analyzed by the relevant National Regulatory Authorities.
This is to emphasize that the abovementioned drug products are not registered with FDA. However, it is important to detect and remove these products from circulation to prevent harm to patients.
Therefore, all Local Government Units (LGU) and Law Enforcement Agencies (LEAs), after the issuance of this advisory, are requested to ensure that these substandard drug products are not sold or not administered to patients in their localities or areas of jurisdiction.
For more information and inquiries, please e-mail us at [email protected]. To report unauthorized sale, or distribution of the abovementioned, kindly e-mail us via [email protected]. You may also call the Center for Drug Regulation and Research at telephone number (02) 8809-5596.
Dissemination of the information to all concerned is highly requested.






