FDA Advisory No. 2020-2105 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Xiao’er Qixingcha Keli 2. Suyingao 20ml 3. Mifepristone […]
FDA Advisory No. 2020-2104-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:
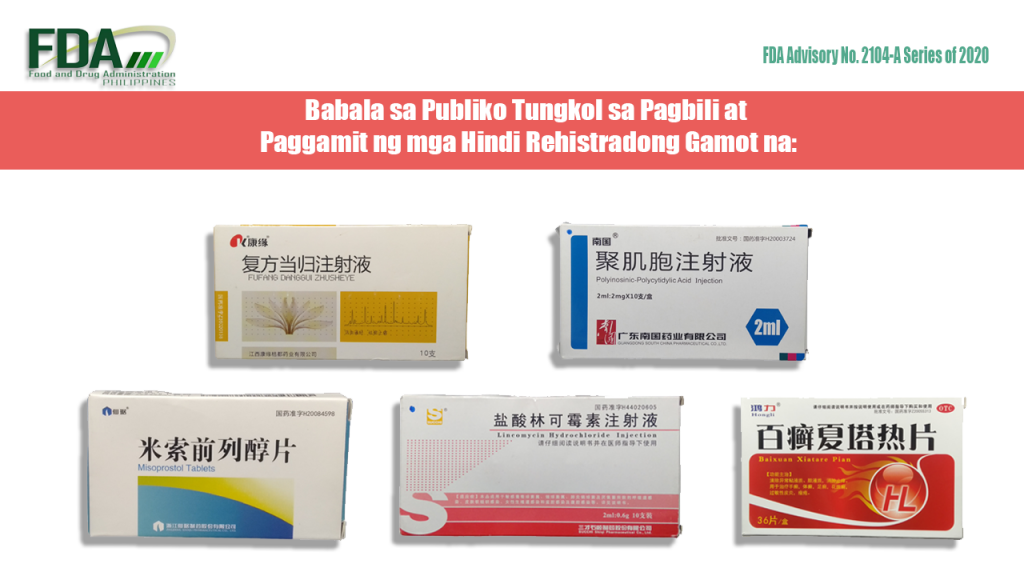
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. K® Fufang Danggui Zhusheye 2ml 2. Misoprostol Tablets 0.2mg 3. Polyinosinic-Polycytidylic […]
FDA Advisory No. 2020-2104 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. K® Fufang Danggui Zhusheye 2ml 2. Misoprostol Tablets 0.2mg […]
FDA Advisory No. 2020-2103-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. OTC Fufang Ganmaoling Pian 2. CSPC® Adenosine Disodium Triphosphate Injection 2ml:20mg […]
FDA Advisory No. 2020-2103 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Fufang Ganmaoling Pian 2. CSPC® Adenosine Disodium Triphosphate […]
FDA Advisory No. 2020-2102-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. Vitamin B12 Injection 1ml:0.5mg 2. Gentamicin Sulfate Injection 2ml:80mg 3. Omeprazole […]
FDA Advisory No. 2020-2102 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Vitamin B12 Injection 1ml:0.5mg 2. Gentamicin Sulfate Injection 2ml:80mg […]
FDA Advisory No. 2020-2101-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. Tylenol® 500 mg tablet [Secondary Packaging] Tylenol® Tab Acetaminophen 500 mg […]
FDA Advisory No. 2020-2101 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Tylenol® 500 mg tablet [Secondary Packaging] Tylenol® Tab Acetaminophen […]
FDA Advisory No. 2020-2100-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Hindi Rehistradong Gamot

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga hindi rehistradong gamot na: 1. OTC Z19991005 [Label in Foreign Language] 2. Zjiachmei® Albumin Human Immunity […]



