FDA Advisory No.2024-0326 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Samil Forus 5g Ophthalmic Ointment 2. Acnon 13 g […]
FDA Advisory No.2024-0325-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. Acetaminophen Capsules Spasmo-Proxyvon® Plus 8’s 2. Baidyanath Asli […]
FDA Advisory No.2024-0325 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Acetaminophen Capsules Spasmo-Proxyvon® Plus 8’s 2. Baidyanath Asli Ayurved […]
FDA Advisory No.2024-0324-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. Jinling Sanshedan Chuanbei Ye 10 mL Vial 12’s […]
FDA Advisory No.2024-0324 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Jinling Sanshedan Chuanbei Ye 10 mL Vial 12’s 2. […]
FDA Advisory No.2024-0323-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. Cefradine Capsules 0.25 g 2. Rengong Niuhuang Jiaxiaozuo […]
FDA Advisory No.2024-0323 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. Cefradine Capsules 0.25 g 2. Rengong Niuhuang Jiaxiaozuo Jiaonang […]
FDA Advisory No.2024-0322-A || Babala sa Publiko tungkol sa Paggamit ng mga sumusunod na Beripikadong Pekeng Gamot na:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na beripikadong pekeng gamot: 1. Ibuprofen + Paracetamol (Alaxan® FR) 200 mg/ 325 […]
FDA Advisory No.2024-0322 || Public Health Warning Against the Purchase and Use of the following Verified Counterfeit Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the counterfeit version of the following products: 1. Ibuprofen + Paracetamol (Alaxan® FR) 200 mg/ […]
FDA Advisory No.2024-0321-A || Babala sa Publiko tungkol sa Paggamit ng mga sumusunod na Beripikadong Pekeng Gamot na:
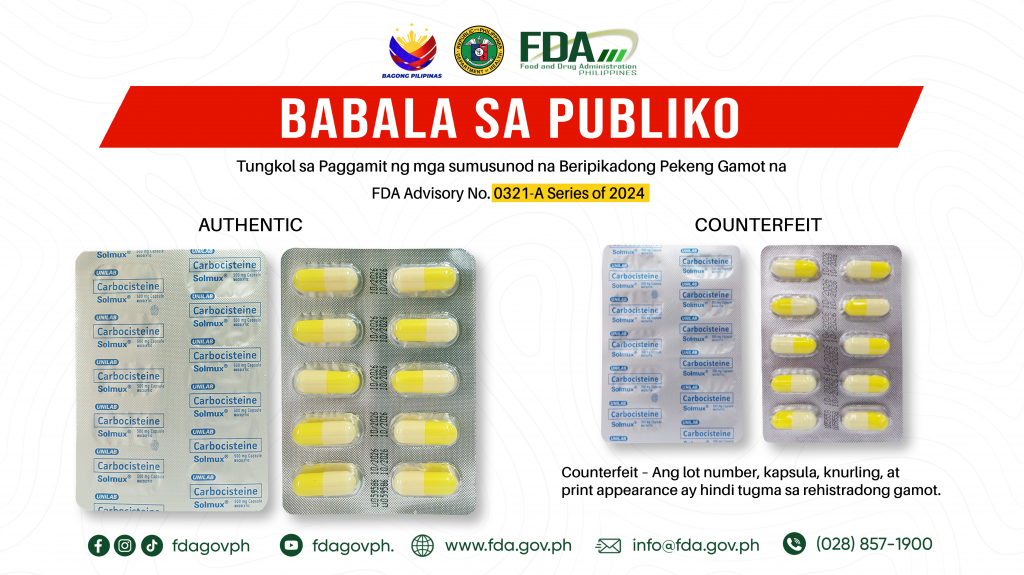
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na beripikadong pekeng gamot: 1. Carbocisteine (Solmux®) 500 mg Capsule 100’s 2. Ibuprofen […]



