FDA Advisory No.2023-2526 || Public Health Warning Against the Purchase and Use of the following Verified Counterfeit Drug Products:
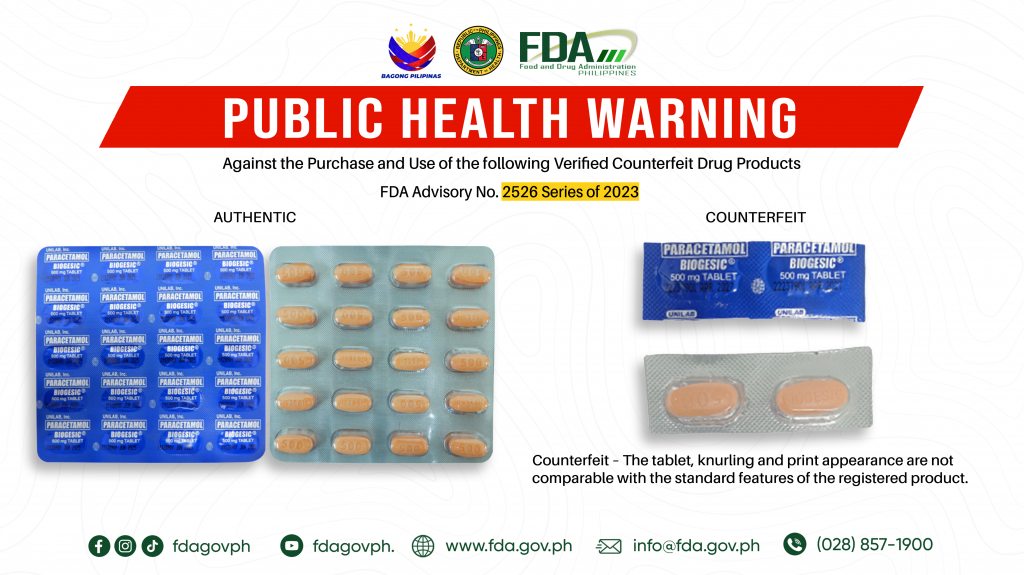
The Food and Drug Administration (FDA) advises the public against the purchase and use of the counterfeit version of the following products: 1. Paracetamol (Biogesic) 500 mg Tablet 2. Ibuprofen […]
FDA Advisory No.2023-2532-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot na “Cefaclor Capsules 0.25 g”
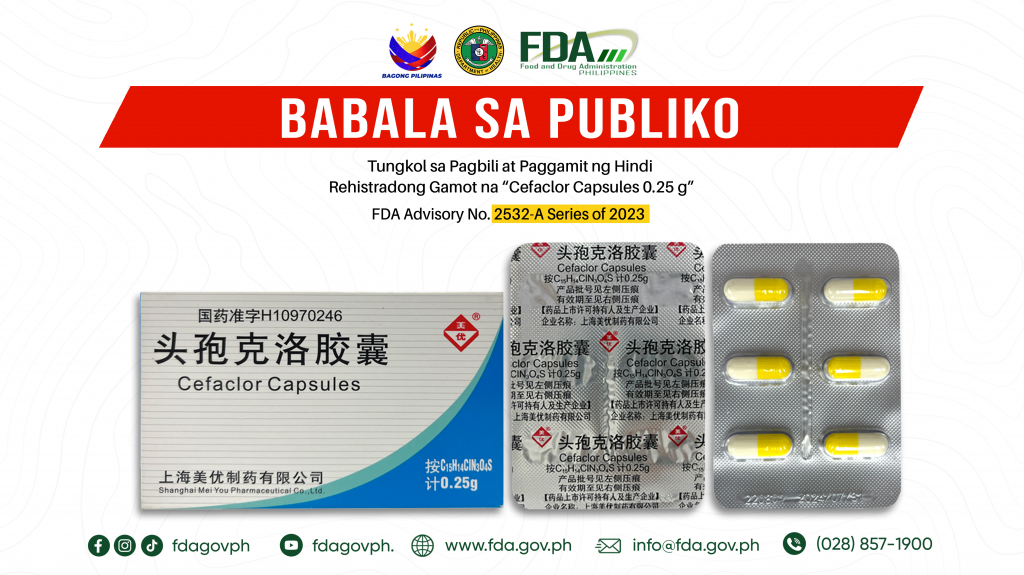
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot na: 1. Cefaclor Capsules 0.25 g Napatunayan sa pamamagitan ng isinagawang Post-Marketing […]
FDA Advisory No.2023-2525-A || Babala sa Publiko tungkol sa Paggamit ng Beripikadong Pekeng Gamot na “Paracetamol + Caffeine (Rexidol® Forte) 500 mg/ 65 mg Tablet”
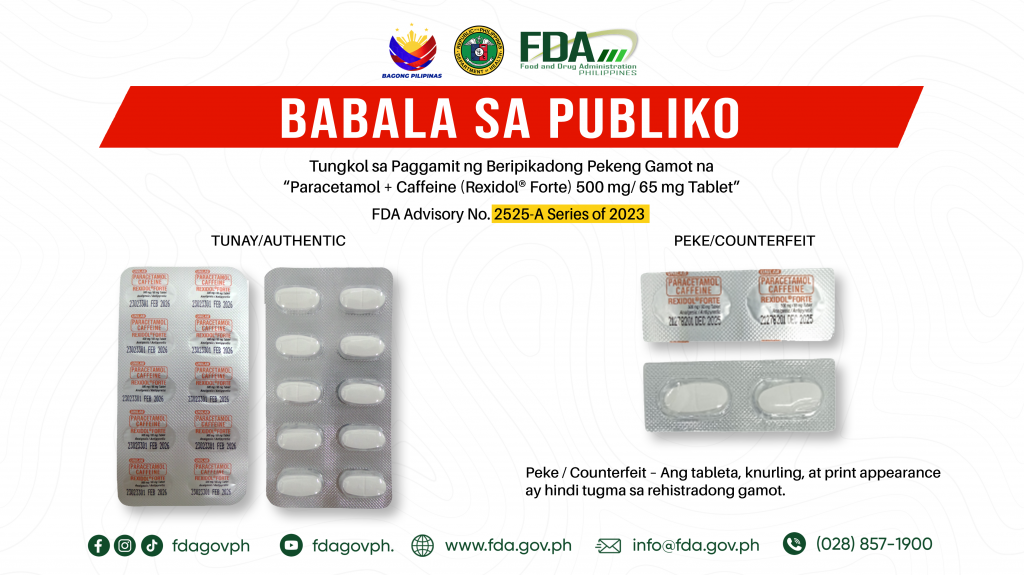
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit nitong sumusunod na beripikadong pekeng gamot: 1. Paracetamol + Caffeine (Rexidol® Forte) 500 mg/ 65 mg […]
FDA Advisory No.2023-2532 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “Cefaclor Capsules 0.25 g” 1.
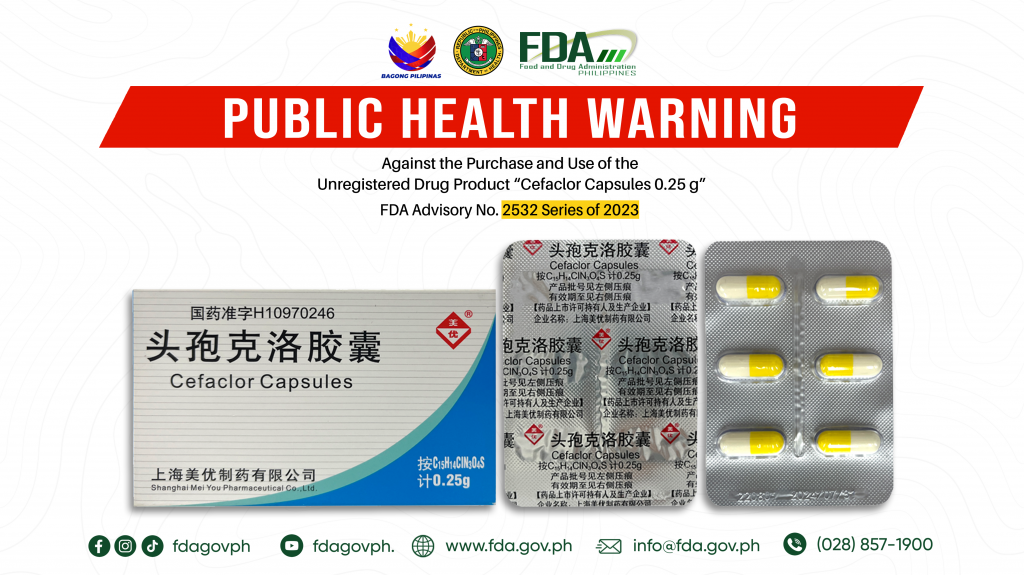
The Food and Drug Administration (FDA) advises the public against the purchase and use of the unregistered drug product: 1. Cefaclor Capsules 0.25 g FDA Post-Marketing Surveillance (PMS) activities have […]
FDA Advisory No.2023-2525 || Public Health Warning Against the Purchase and Use of the Verified Counterfeit Drug Product “Paracetamol + Caffeine (Rexidol® Forte) 500 mg/ 65 mg Tablet”
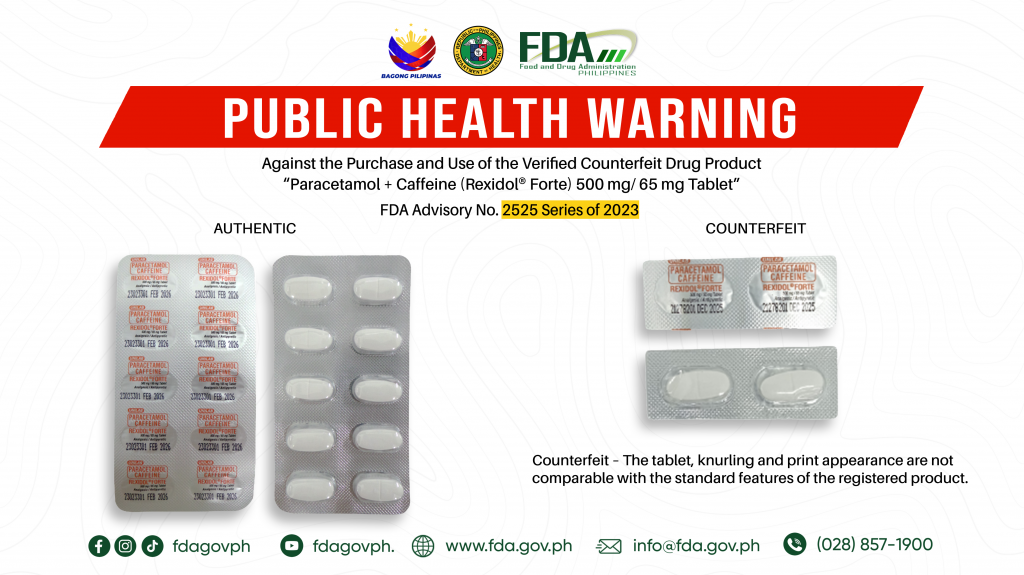
The Food and Drug Administration (FDA) advises the public against the purchase and use of the counterfeit version of the following product: 1. Paracetamol + Caffeine (Rexidol® Forte) 500 mg/ […]
FDA Advisory No.2023-2531-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot na “OTC Keteling Jiaonang [As reflected in the package insert]”

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot na: 1. OTC Keteling Jiaonang [As reflected in the package insert] Napatunayan […]
FDA Advisory No.2023-2531 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “OTC Keteling Jiaonang [As reflected in the package insert]”

The Food and Drug Administration (FDA) advises the public against the purchase and use of the unregistered drug product: 1. OTC Keteling Jiaonang [As reflected in the package insert] FDA […]
FDA Advisory No.2023-2530-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot na “OTC SUNFLOWER Xiao’er Chaigui Tuire Keli [As reflected in the package insert]”

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot na: 1. OTC SUNFLOWER Xiao’er Chaigui Tuire Keli [As reflected in the […]
FDA Advisory No.2023-2530 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “OTC SUNFLOWER Xiao’er Chaigui Tuire Keli [As reflected in the package insert]”

The Food and Drug Administration (FDA) advises the public against the purchase and use of the unregistered drug product: 1. OTC SUNFLOWER Xiao’er Chaigui Tuire Keli [As reflected in the […]
FDA Advisory No.2023-2529-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot na “Dahongying Meloxicam Tablets 7.5 mg”

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot na: 1. Dahongying Meloxicam Tablets 7.5 mg Napatunayan sa pamamagitan ng isinagawang […]



