FDA Advisory No.2024-0748 || Pilot Implementation of the Electronic Certificate of Product Registration (E-CPR) Information System for the Application for Initial Registration of Generic Prescription Drugs for Human Use from 07 May 2024 to 07 August 2024

The Food and Drug Administration (FDA) has developed the Electronic Certificate of Product Registration (e-CPR) Information System, which shall be included in the FDA eServices Portal System and shall serve […]
FDA Advisory No.2024-0573 || Call for Nomination as member of the Animal Health Product Advisory Committee (AHPAC)

Pursuant to Republic Act No. 9711, otherwise known as the “Food and Drug Administration (FDA) Act of 2009”, the FDA is responsible for the issuance of appropriate authorizations, such as […]
FDA Advisory No.2024-0558 || Call for Nomination as member of the Human Drug Registration Advisory Committee (HDRAC)

Pursuant to Republic Act No. 9711, otherwise known as the “Food and Drug Administration (FDA) Act of 2009”, the FDA is responsible for the issuance of appropriate authorizations, such as […]
FDA Advisory No.2024-0747-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot “OTC Erythromycin Ointment 5g”
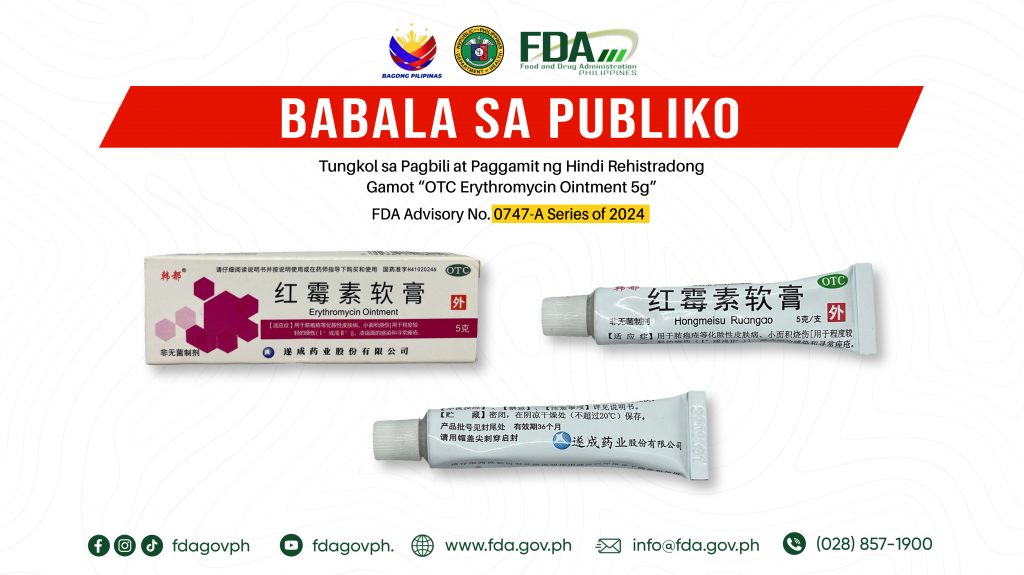
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot: 1. OTC Erythromycin Ointment 5g Napatunayan sa pamamagitan ng isinagawang Post-Marketing Surveillance […]
FDA Advisory No.2024-0747 || Public Health Warning Against the Purchase and Use of the Unregistered Drug Product “OTC Erythromycin Ointment 5g”
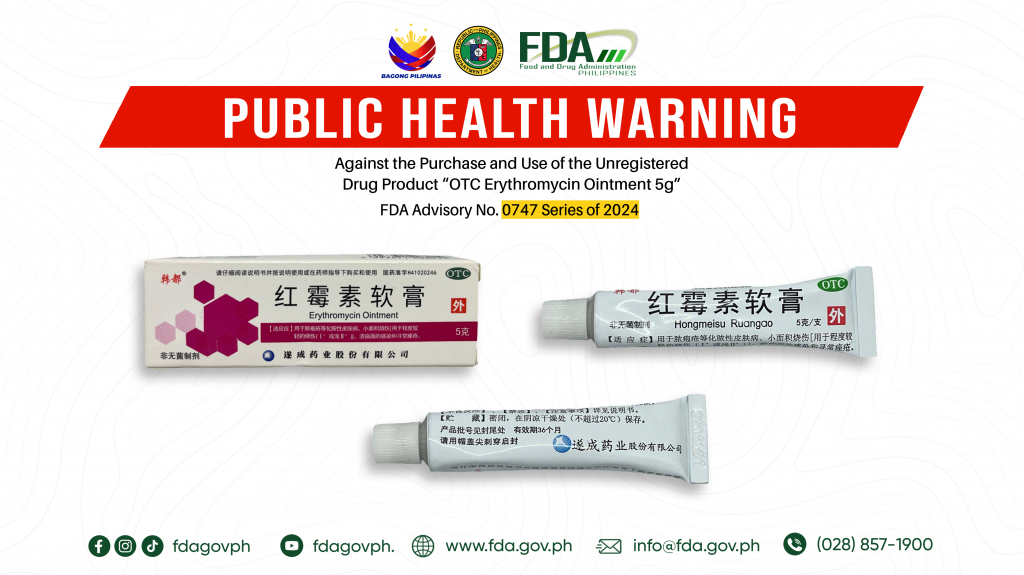
The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug product: 1. OTC Erythromycin Ointment 5g FDA Post-Marketing Surveillance (PMS) activities […]
FDA Advisory No.2024-0746-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. OTC Topfond Luleitading Pian Loratadine Tablets 2. Lihua […]
FDA Advisory No.2024-0746 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:

The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Topfond Luleitading Pian Loratadine Tablets 2. Lihua Bidouyan […]
FDA Advisory No.2024-0745-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng mga Sumusunod na Hindi Rehistradong Gamot:
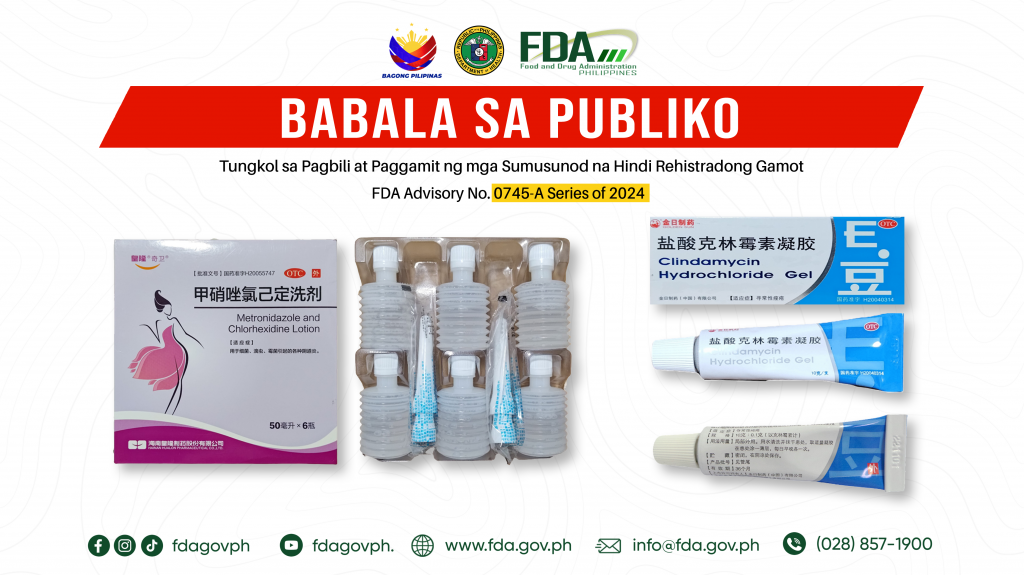
Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng mga sumusunod na hindi rehistradong gamot: 1. OTC Metronidazole and Chlorhexidine Lotion 2. OTC GOLDEN […]
FDA Advisory No.2024-0745 || Public Health Warning Against the Purchase and Use of the Following Unregistered Drug Products:
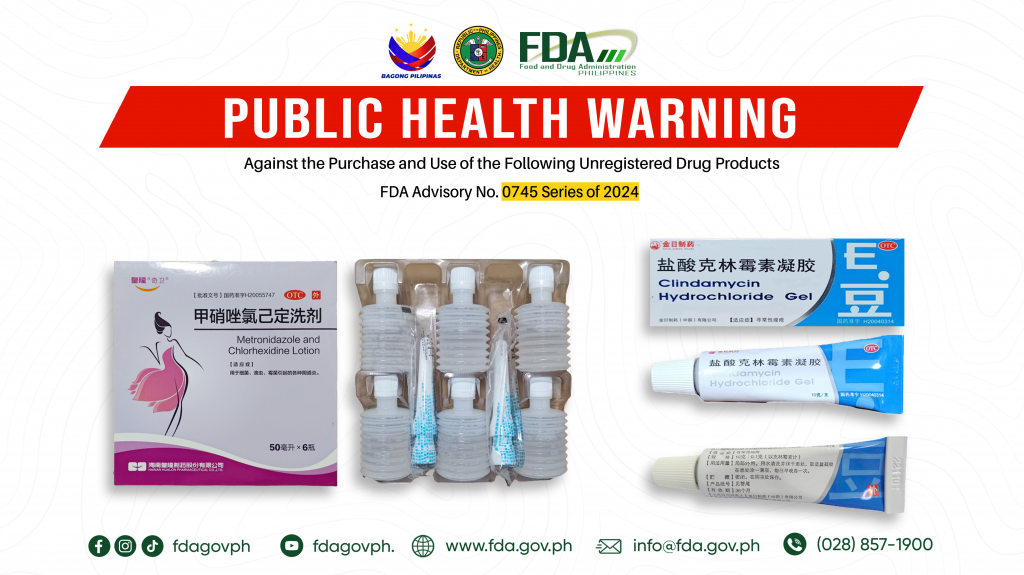
The Food and Drug Administration (FDA) advises the public against the purchase and use of the following unregistered drug products: 1. OTC Metronidazole and Chlorhexidine Lotion 2. OTC GOLDEN SUN […]
FDA Advisory No.2024-0744-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot “NANZ i-Drops 15ml”

Pinapayuhan ng Food and Drug Administration (FDA) ang publiko laban sa pagbili at paggamit ng hindi rehistradong gamot: 1. NANZ i-Drops 15ml Napatunayan sa pamamagitan ng isinagawang Post-Marketing Surveillance (PMS) […]



